Exercise preconditioning mitigates brain injury after cerebral ischemia-reperfusion injury in rats by restraining TIMP1
- PMID: 39364701
- PMCID: PMC11450454
- DOI: 10.1002/iid3.70008
Exercise preconditioning mitigates brain injury after cerebral ischemia-reperfusion injury in rats by restraining TIMP1
Abstract
Background: Cerebral ischemic disease is a common cerebrovascular disease, especially ischemic stroke. Exercise has protective functions on brain tissues following cerebral ischemia-reperfusion injury (CIRI), but its preventive effects and mechanisms in CIRI remain unclear. We aimed to investigate the effects and mechanisms of exercise preconditioning on CIRI.
Methods: The middle cerebral artery occlusion (MCAO) operation was prepared to establish CIRI rats. All rats were randomized into the MCAO, exercise (exercise preconditioning plus MCAO operation), vector (exercise preconditioning, MCAO operation plus intraventricular injection of empty vector), and tissue inhibitor of metalloprotease 1 overexpression (OE-TIMP1, exercise preconditioning, MCAO operation plus intraventricular injection of OE-TIMP1) groups.
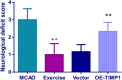
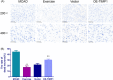
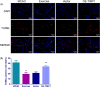
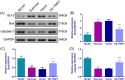
Results: The results indicated that exercise preconditioning suppressed approximately 66.67% of neurological deficit scores and 73.79% of TIMP1 mRNA expression in MCAO rats, which were partially offset by OE-TIMP1. The protective effects of exercise against neuron death status and cerebral infarction size in MCAO rats were reversed by OE-TIMP1. It also confirmed that exercise weakened apoptosis and oxidative stress damage, with notable increases of B-cell lymphoma-2, superoxide dismutase, and glutathione peroxidase production, and evident decreases of BCL2-associated X, caspase 3, and malondialdehyde in MCAO rats, while these effects were partially reversed by OE-TIMP1. Additionally, the inhibitory effects of exercise on the protein levels of TIMP1, hypoxia-inducible factor-alpha, vascular endothelial growth factor receptor 2, vascular endothelial growth factor, and neurogenic locus notch homolog protein 1 in MCAO rats were partially reversed by OE-TIMP1.
Conclusion: Altogether, exercise preconditioning had protective effects on CIRI by restraining TIMP1, which provided new therapeutic strategies for preventing CIRI.
Keywords: TIMP1; brain injury; cerebral ischemia‐reperfusion injury; exercise preconditioning.
© 2024 The Author(s). Immunity, Inflammation and Disease published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
RNA sequencing reveals the potential mechanism of exercise preconditioning for cerebral ischemia reperfusion injury in rats.Brain Behav. 2024 Jul;14(7):e3608. doi: 10.1002/brb3.3608. Brain Behav. 2024. PMID: 38956886 Free PMC article.
-
Role of the Janus kinase 2/signal transducers and activators of transcription 3 pathway in the protective effect of remote ischemia preconditioning against cerebral ischemia-reperfusion injury in rats.Neuroreport. 2019 Jun 12;30(9):664-670. doi: 10.1097/WNR.0000000000001257. Neuroreport. 2019. PMID: 30969244 Free PMC article.
-
Exercise preconditioning increases circulating exosome miR-124 expression and alleviates apoptosis in rats with cerebral ischemia-reperfusion injury.Brain Res. 2025 Mar 15;1851:149457. doi: 10.1016/j.brainres.2025.149457. Epub 2025 Jan 15. Brain Res. 2025. PMID: 39824375
-
HACE1 exerts a neuroprotective role against oxidative stress in cerebral ischemia-reperfusion injury by activating the PI3K/AKT/Nrf2 pathway.Neuroscience. 2024 Nov 1;559:249-262. doi: 10.1016/j.neuroscience.2024.09.002. Epub 2024 Sep 6. Neuroscience. 2024. PMID: 39244008
-
Mechanisms Associated with Mitophagy and Ferroptosis in Cerebral Ischemia-reperfusion Injury.J Integr Neurosci. 2025 Mar 18;24(3):26458. doi: 10.31083/JIN26458. J Integr Neurosci. 2025. PMID: 40152564 Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

