Self-assembling nanoparticle engineered from the ferritinophagy complex as a rabies virus vaccine candidate
- PMID: 39366932
- PMCID: PMC11452399
- DOI: 10.1038/s41467-024-52908-z
Self-assembling nanoparticle engineered from the ferritinophagy complex as a rabies virus vaccine candidate
Abstract
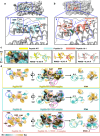
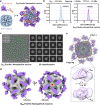
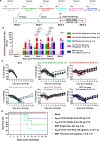
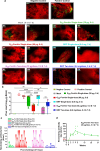
Over the past decade, there has been a growing interest in ferritin-based vaccines due to their enhanced antigen immunogenicity and favorable safety profiles, with several vaccine candidates targeting various pathogens advancing to phase I clinical trials. Nevertheless, challenges associated with particle heterogeneity, improper assembly and unanticipated immunogenicity due to the bulky protein adaptor have impeded further advancement. To overcome these challenges, we devise a universal ferritin-adaptor delivery platform based on structural insights derived from the natural ferritinophagy complex of the human ferritin heavy chain (FTH1) and the nuclear receptor coactivator 4 (NCOA4). The engineered ferritinophagy (Fagy)-tag peptide demonstrate significantly enhanced binding affinity to the 24-mer ferritin nanoparticle, enabling efficient antigen presentation. Subsequently, we construct a self-assembling rabies virus (RABV) vaccine candidate by noncovalently conjugating the Fagy-tagged glycoprotein domain III (GDIII) of RABV to the ferritin nanoparticle, maintaining superior homogeneity, stability and immunogenicity. This vaccine candidate induces potent, rapid, and durable immune responses, and protects female mice against the authentic RABV challenge after single-dose administration. Furthermore, this universal, ferritin-based antigen conjugating strategy offers significant potential for developing vaccine against diverse pathogens and diseases.
© 2024. The Author(s).
Conflict of interest statement
Y.G., D.F., and P.Y. declare the following competing interests: patent has been filed for the Fagy-tag and self-assembly nanoparticle system presented here. All other authors declare no competing interests.
Figures



References
-
- Munro, H. N. & Linder, M. C. Ferritin: structure, biosynthesis, and role in iron metabolism. Physiol. Rev.58, 317–396 (1978). - PubMed
-
- Harrison, P. M. & Arosio, P. The ferritins: molecular properties, iron storage function and cellular regulation. Biochim. Biophys. Acta (BBA) Bioenerg.1275, 161–203 (1996). - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

