Oropharyngeal Adverse Events to Drugs and Vaccines: Pharmacovigilance Data From Italy (2019-2021)
- PMID: 39370676
- PMCID: PMC12021317
- DOI: 10.1111/odi.15145
Oropharyngeal Adverse Events to Drugs and Vaccines: Pharmacovigilance Data From Italy (2019-2021)
Abstract
Objectives: The aim of this study was to perform a descriptive analysis of oropharyngeal adverse events (AEs) related to drugs and/or vaccines in order to provide useful information for clinicians.
Methods: Data related to three regions of Italy were analyzed from 2019 to 2021 by using the National Pharmacovigilance database.
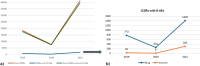
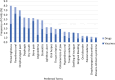
Results: Among overall 67,384 cases, 2773 (4.1%) reported at least one oropharyngeal AE. Most cases referred to females (71.0%) and adults (70.8%). The majority of cases were reported as not serious (68.4%) and the outcome was mainly positive (73.5%). The cases related to drugs (52.2%) were slightly more than those related to vaccines (47.8%), the latter nearly completely represented by COVID-19 vaccines. Among 3324 oropharyngeal AEs, the most commonly reported were oropharyngeal conditions (65.9%). The most reported AEs related to vaccines were paresthesia oral and oropharyngeal pain, whereas the most reported AEs related to drugs were throat tightness and angioedema. A marked under-reporting of osteonecrosis of the jaw (2.9%) was observed, despite this risk was well documented in the same country.
Conclusions: This analysis suggested an under-reporting of oropharyngeal AEs and the need to better train dentists, dental hygienists, and also general practitioners.
Keywords: database; dentists; oropharyngeal adverse events; pharmacovigilance; spontaneous reporting.
© 2024 The Author(s). Oral Diseases published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

