Creating a suprazyme: integrating a molecular enzyme mimic with a nanozyme for enhanced catalysis
- PMID: 39371455
- PMCID: PMC11450938
- DOI: 10.1039/d4sc04577g
Creating a suprazyme: integrating a molecular enzyme mimic with a nanozyme for enhanced catalysis
Abstract
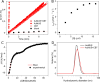
Enzyme mimics, due to their limited complexity, traditionally display low catalytic efficiency. Herein we present a strategy that enables the transformation of a slow-acting catalyst into a highly active one by creating a non-covalent suprastructure, termed "suprazyme". We show that cucurbit[7]uril macrocycles, rudimentary molecular enzyme mimics, embedded within an anionic monolayer on the surface of gold nanoparticles, outperform individual cucurbit[7]urils as well as nanoparticles, which also exhibit catalytic enzyme-like activity and thus act as nanozymes, by over 50 times, showcasing a 1044-fold acceleration in a model oxime formation reaction. The superior performance of such a suprazyme is attributed to a synergistic interplay between the organic monolayer and macrocycles, which is accompanied by a decreased local polarity and pH that favors the acid-catalyzed condensation process. The proposed approach holds promise for developing diverse suprazymes, contingent upon achieving a complementary structure and mechanism of action between the molecular catalyst and nanoparticles.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures



References
LinkOut - more resources
Full Text Sources

