ACKR3 agonism induces heterodimerization with chemokine receptor CXCR4 and attenuates platelet function
- PMID: 39373210
- PMCID: PMC11628653
- DOI: 10.1111/eci.14327
ACKR3 agonism induces heterodimerization with chemokine receptor CXCR4 and attenuates platelet function
Abstract
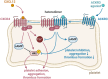
Background: Platelet receptors ACKR3 and CXCR4 play a crucial role in a variety of cardiovascular diseases. Like most chemokine receptors, CXCR4 is a G protein coupled receptor that induces platelet activation. In contrast, the atypical chemokine receptor 3 (ACKR3) lacks the ability to activate heterotrimeric G proteins and its activation leads to platelet inhibition and attenuates thrombus formation. In nucleated cells, heterodimerization of ACKR3 with CXCR4 regulates CXCL12-dependent signalling. The aim of our study was to investigate the formation of ACKR3/CXCR4 heterodimers in platelets and the subsequent consequences for platelet function.
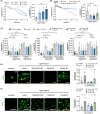
Methods and results: Using a proximity ligation assay (PLA, Duolink®) to screen for CXCR4/ACKR3 heterodimerization inducing compounds, we found that ACKR3 agonism but not conventional platelet agonists or endogen ligands lead to heterodimer formation. To further characterize the formation of ACKR3/CXCR4 heterodimers, we studied the CXCL12-dependent platelet activation via CXCR4. Both, CXCL12-dependent platelet aggregation and collagen-dependent ex vivo thrombus formation were significantly downregulated by ACKR3 agonism. Moreover, platelet intracellular calcium and Akt signalling were increased by CXCL12 and again suppressed by ACKR3-specific agonists. Previously, CXCL12 was shown to decrease platelet cAMP levels via CXCR4. Treatment with a specific ACKR3 agonist counteracted this CXCL12/CXCR4-dependent cAMP decrease.
Conclusion: Our results reveal that the formation of platelet ACKR3/CXCR4 heterodimers is dependent on ACKR3 rather than CXCR4. Furthermore, ACKR3 agonism induced heterodimerization is associated with mitigating CXCL12/CXCR4-dependent platelet activation possibly by modulating CXCR4-dependent G protein signalling. Our results indicate possible ACKR3 agonist functions and reinforce the potential therapeutic applications of ACKR3 agonists.
Keywords: C‐X‐C motif chemokine ligand 12; C‐X‐C motif chemokine receptor type 4; atypical chemokine receptor 3; heterodimerization; platelet.
© 2024 The Author(s). European Journal of Clinical Investigation published by John Wiley & Sons Ltd on behalf of Stichting European Society for Clinical Investigation Journal Foundation.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures


References
-
- Clemetson KJ, Clemetson JM, Proudfoot AE, Power CA, Baggiolini M, Wells TN. Functional expression of CCR1, CCR3, CCR4, and CXCR4 chemokine receptors on human platelets. Blood. 2000;96(13):4046‐4054. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

