Validation of Roche immunoassay for severe acute respiratory coronavirus 2 in South Africa
- PMID: 39376942
- PMCID: PMC11457558
- DOI: 10.4102/sajid.v36i1.286
Validation of Roche immunoassay for severe acute respiratory coronavirus 2 in South Africa
Abstract
Background: Serology testing is an important ancillary diagnostic to the reverse transcriptase polymerase chain reaction (RT-PCR) test for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). We aimed to evaluate the performance of the Roche Elecsys™ chemiluminescent immunoassay (Rotkreuz, Switzerland), that detects antibodies against the SARS-CoV-2 nucleocapsid antigen, at an academic laboratory in South Africa.
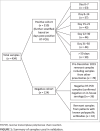
Methods: Serum samples were collected from 312 donors with confirmed positive SARS-CoV-2 RT-PCR tests, with approval from a large university's human research ethics committee. Negative controls included samples stored prior to December 2019 and from patients who tested negative for SARS-CoV-2 on RT-PCR and were confirmed negative using multiple serology methods (n = 124). Samples were stored at -80 °C and analysed on a Roche cobas™ 602 autoanalyser.
Results: Compared with RT-PCR, our evaluation revealed a specificity of 100% and overall sensitivity of 65.1%. The sensitivity in individuals > 14 days' post-diagnosis was 72.6%, with the highest sensitivity 31-50 days' post-diagnosis at 88.6%. Results were also compared with in-house serology tests that showed high agreement in majority of categories.
Conclusions: The sensitivity at all-time points post-diagnosis was lower than reported in other studies, but sensitivity in appropriate cohorts approached 90% with a high specificity. The lower sensitivity at earlier time points or in individuals without symptomatology may indicate failure to produce antibodies, which was further supported by the comparison against in-house serology tests.
Keywords: COVID-19; SARS-CoV-2; antibodies; immunoglobulin G; immunoglobulin M; serology; validation.
© 2021. The Authors.
Conflict of interest statement
The authors declare that they have no financial or personal relationships that may have inappropriately influenced them in writing this article.
Figures
References
-
- World Health Organization . Weekly operational update on COVID-19 [homepage on the Internet]. [cited 2021 Mar 29] p. 1–10. Available from: https://www.who.int/publications/m/item/weekly-operational-update-on-cov...
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous