Design of PtSn Nanocatalysts for Fuel Cell Applications
- PMID: 39382180
- PMCID: PMC11639638
- DOI: 10.1002/cplu.202400151
Design of PtSn Nanocatalysts for Fuel Cell Applications
Abstract
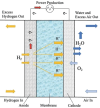
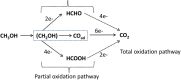
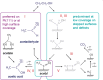
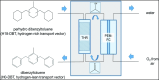
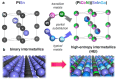
The challenges in the fuel cell industry lie in the cost, performance, and durability of the electrode components, especially the platinum-based catalysts. Alloying has been identified as an effective strategy to reduce the cost of the catalyst and increase its efficiency and durability. So far, most studies focused on the design of PtM bimetallic nanocatalyst, where M is a transition metal. The resulting PtM materials show higher catalytic activity, but their stability remained challenging. In addition, most of the transition metals M are expensive or low abundant. Tin (Sn) has gained attention as alloying element due to its versatility in manufacturing both anode and cathode electrodes. If used as anode catalyst, it is able to overcome poisoning from CO and related intermediates. As cathode catalyst, it improves the kinetics of the oxygen reduction reaction (ORR). Additionally, Sn is an abundant and cheap element. The current contribution outlines the state of the art on the alloy and shape effect on PtSn activity and stability, demonstrating its high potential to develop cheaper, more efficient and durable catalysts for fuel-cell electrodes. Additionally, in situ analytical and spectroscopic studies can shed light on the elementary steps involved in the use of PtSn catalytic systems. Finally, this intriguing material can be used as a parent system for the synthesis of high-entropy-alloys and intermetallics materials.
Keywords: Alloys; Intermetallics; Mobility; Nanoparticles; sustainability.
© 2024 The Authors. ChemPlusChem published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
-
- K. K. Karuppanan, M. K. Panthalingal, P. Biji, Handbook of Nanomaterials for Industrial Applications, Elsevier, 2018, 468–495.
-
- Borup R. L., Ahmet Kusoglu, Neyerlin K. C., Ahluwalia R. M. R. K., Cullen D. A., More K. L., Weber A. Z., Myers D. J., Curr. Opin. Electroche 2020, 21, 192–200.
-
- World Platinum Investment Council, Fuel cell electric vehicles and platinum demand, https://www.cmegroup.com/articles/2022/wpic-fuel-cell-electric-vehicles-..., (accessed on February 2024).
-
- Yan J., Jing J., Li Y., Int. J. Hydrog. Energy 2024, 52, 734–749.
-
- Ghosh P., Clean Technologies and Environmental Policies 2017, 19, 595–601.
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

