Case report: MEK inhibitor as treatment for multi-lineage mosaic KRAS G12D-associated epidermal nevus syndrome in a pediatric patient
- PMID: 39385823
- PMCID: PMC11461199
- DOI: 10.3389/fneur.2024.1466946
Case report: MEK inhibitor as treatment for multi-lineage mosaic KRAS G12D-associated epidermal nevus syndrome in a pediatric patient
Abstract
The RASopathies, collectively, are a spectrum of genetic syndromes caused by mutations in genes involved in the RAS/ mitogen-activated protein kinase (MAPK) pathway, including but not limited to PTPN11, NRAS, KRAS, HRAS, BRAF, and MAP2K1. Recognized RASopathy conditions include neurofibromatosis type 1 (NF1), Noonan syndrome, capillary malformation-arteriovenous malformation syndrome, Costello syndrome, cardiofacio-cutaneous (CFC) syndrome, LEOPARD syndrome and Legius syndrome. The RASopathies often display overlapping clinical features, presumably owing to common RAS-MAPK signaling pathway activation driving dysregulated cell proliferation. Epidermal nevus syndromes (ENS) are described as the presence of epidermal nevi, in individuals also affected by extra-cutaneous organ system involvement, and there is recent recognition of mosaic RAS mutations as molecular drivers of ENS. Currently, no curative treatments exist for RASopathy driven conditions, but rather symptom-directed management is the currently accepted standard. Here, we detail a unique case of a child exhibiting diffuse spinal nerve root hypertrophy in the context of epidermal nevus syndrome driven by molecularly confirmed KRAS G12D mosaicism, treated with the MEK 1/2 inhibitor selumetinib. Herein, we report the response of this patient to targeted therapy of more than two years' duration, including stabilization of multilevel nerve root hypertrophy as well as significant improvement in epidermal nevi. While the effectiveness of MEK inhibitors such as selumetinib is established in NF1-associated inoperable plexiform neurofibromas, their use in managing hyperactive KRAS-driven epidermal nevi and hypertrophic neuropathy remains unproven, and this case, to our knowledge, is the first such case to be reported. Shared molecular dysregulation and overlapping clinical features between these conditions suggest potential for effective therapeutic application of MEK directed therapy to address a range of conditions resulting from germline and/ or mosaic expression of aberrantly regulated RAS signaling.
Keywords: KRAS G12D; RASopathy; epidermal nevus syndrome; hypertrophic neuropathy; selumetinib.
Copyright © 2024 Dionysiou, Makri, Ahlawat, Guryildirim, Barañano, Groves, Argani and Pratilas.
Conflict of interest statement
C.A.P. has received research grants from Kura Oncology and Novartis Institutes for Biomedical Research (unrelated to this manuscript); and consulting fees from Day One Therapeutics (unrelated). The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


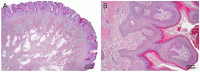

References
-
- Beyens A, Dequeker L, Brems H, Janssens S, Syryn H, D’Hooghe A, et al. Identification of codon 146 variants in isolated epidermal nevus and multiple lesions in oculoectodermal syndrome: confirmation of the phenotypic continuum of mosaic RASopathies. Int J Mol Sci. (2022) 23:74036. doi: 10.3390/ijms23074036, PMID: - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

