Immune Implications of Cholesterol-Containing Lipid Nanoparticles
- PMID: 39388645
- PMCID: PMC11505898
- DOI: 10.1021/acsnano.4c06369
Immune Implications of Cholesterol-Containing Lipid Nanoparticles
Abstract
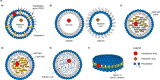
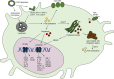
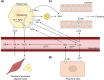
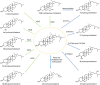
The majority of clinically approved nanoparticle-mediated therapeutics are lipid nanoparticles (LNPs), and most of these LNPs are liposomes containing cholesterol. LNP formulations significantly alter the drug pharmacokinetics (PK) due to the propensity of nanoparticles for uptake by macrophages. In addition to readily engulfing LNPs, the high expression of cholesterol hydroxylases and reactive oxygen species (ROS) in macrophages suggests that they will readily produce oxysterols from LNP-associated cholesterol. Oxysterols are a heterogeneous group of cholesterol oxidation products that have potent immune modulatory effects. Oxysterols are implicated in the pathogenesis of atherosclerosis and certain malignancies; they have also been found in commercial liposome preparations. Yet, the in vivo metabolic fate of LNP-associated cholesterol remains unclear. We review herein the mechanisms of cellular uptake, trafficking, metabolism, and immune modulation of endogenous nanometer-sized cholesterol particles (i.e., lipoproteins) that are also relevant for cholesterol-containing nanoparticles. We believe that it would be imperative to better understand the in vivo metabolic fate of LNP-associated cholesterol and the immune implications for LNP-therapeutics. We highlight critical knowledge gaps that we believe need to be addressed in order to develop safer and more efficacious lipid nanoparticle delivery systems.
Keywords: cholesterol; cholesterol oxidation; immune modulation; lipid metabolism; lipid nanoparticle; liposome; macrophage; oxysterol.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

