Current Overview of the Biology and Pharmacology in Sugen/Hypoxia-Induced Pulmonary Hypertension in Rats
- PMID: 39388691
- PMCID: PMC11502635
- DOI: 10.1089/jamp.2024.0016
Current Overview of the Biology and Pharmacology in Sugen/Hypoxia-Induced Pulmonary Hypertension in Rats
Abstract
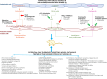
The Sugen 5416/hypoxia (Su/Hx) rat model of pulmonary arterial hypertension (PAH) demonstrates most of the distinguishing features of PAH in humans, including increased wall thickness and obstruction of the small pulmonary arteries along with plexiform lesion formation. Recently, significant advancement has been made describing the epidemiology, genomics, biochemistry, physiology, and pharmacology in Su/Hx challenge in rats. For example, there are differences in the overall reactivity to Su/Hx challenge in different rat strains and only female rats respond to estrogen treatments. These conditions are also encountered in human subjects with PAH. Also, there is a good translation in both the biochemical and metabolic pathways in the pulmonary vasculature and right heart between Su/Hx rats and humans, particularly during the transition from the adaptive to the nonadaptive phase of right heart failure. Noninvasive techniques such as echocardiography and magnetic resonance imaging have recently been used to evaluate the progression of the pulmonary vascular and cardiac hemodynamics, which are important parameters to monitor the efficacy of drug treatment over time. From a pharmacological perspective, most of the compounds approved clinically for the treatment of PAH are efficacious in Su/Hx rats. Several compounds that show efficacy in Su/Hx rats have advanced into phase II/phase III studies in humans with positive results. Results from these drug trials, if successful, will provide additional treatment options for patients with PAH and will also further validate the excellent translation that currently exists between Su/Hx rats and the human PAH condition.
Keywords: PAH pathogenesis; Sugen 5416/hypoxia rat model; current and novel therapies for PAH; pulmonary arterial pressure (PAH); translation between the Su/Hx rat model and human PAH.
Figures

References
-
- Galie N, Humbert M, Vachiery JL, et al. ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension: The joint task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J 2015;46(4):903–975; doi: 10.1183/13993003 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
