Fotemustine in recurrent high‑grade glioma: MRI neuro‑radiological findings
- PMID: 39390978
- PMCID: PMC11465436
- DOI: 10.3892/ol.2024.14703
Fotemustine in recurrent high‑grade glioma: MRI neuro‑radiological findings
Abstract
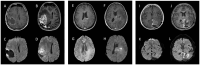
The use of fotemustine (FTM) has been authorized in certain countries for the treatment of recurrent high-grade gliomas (HGG) after Stupp therapy. However, to the best of our knowledge, no studies have assessed changes in magnetic resonance imaging (MRI) during treatment with FTM monotherapy. The aim of the present study was to assess the neuroradiological findings in a cohort of patients with recurrent HGG treated with FTM monotherapy. Patients with HGG already undergoing the Stupp protocol were retrospectively included. MRIs (pre- and post-FTM treatment) were analyzed by two neuroradiologists in consensus: Volume and diffusion values of the contrast-enhanced component were measured on T1-weighted volumetric sequences after gadolinium injection and on apparent diffusion coefficient (ADC) maps, respectively. A total of 19 patients [median age, 49 years; interquartile range (IQR), 43-57 years] were included, 17 of whom had glioblastoma and 2 had astrocytoma isocitrate dehydrogenase-mutated grade 4. The median duration of FTM therapy was 4 months (IQR, 2-6 months). The median tumor volume measured on the contrast-enhanced component was 2,216 mm3 (IQR, 768-13,169 mm3) at baseline and 9,217 mm3 (IQR, 3,455-16,697 mm3) at the end of treatment, with a median change of +38% (IQR, -45-+574%). A total of seven patients showed a volume decrease. ADC value analysis of the enhancement area demonstrated no significant difference between the pre- and the post-FTM treatment periods (P=0.36); however, in three patients, the decreases in ADC levels were particularly marked. In conclusion, the present study described a series of patients with recurrent HGG treated with FTM in monotherapy, demonstrating a prevalent increase in lesion enhancement and three cases of marked restrictions on diffusion-weighted imaging. Further prospective studies are required to corroborate such preliminary results.
Keywords: MRI; apparent diffusion coefficient; diffusion-weighted imaging; fotemustine; glioblastoma; high-grade glioma; recurrence.
Copyright: © Savoldi et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- van den Bent MJ, Tesileanu CMS, Wick W, Sanson M, Brandes AA, Clement PM, Erridge S, Vogelbaum MA, Nowak AK, Baurain JF, et al. Adjuvant and concurrent temozolomide for 1p/19q non-co-deleted anaplastic glioma (CATNON; EORTC study 26053-22054): Second interim analysis of a randomised, open-label, phase 3 study. Lancet Oncol. 2021;22:813–823. doi: 10.1016/S1470-2045(21)00090-5. - DOI - PMC - PubMed
