Elevated levels of damage-associated molecular patterns HMGB1 and S100A8/A9 coupled with toll-like receptor-triggered monocyte activation are associated with inflammation in patients with myelofibrosis
- PMID: 39391311
- PMCID: PMC11465240
- DOI: 10.3389/fimmu.2024.1365015
Elevated levels of damage-associated molecular patterns HMGB1 and S100A8/A9 coupled with toll-like receptor-triggered monocyte activation are associated with inflammation in patients with myelofibrosis
Abstract
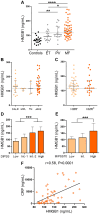
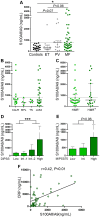
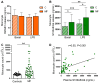
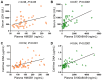
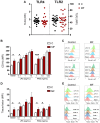
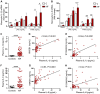
Inflammation plays a pivotal role in the pathogenesis of primary and post-essential thrombocythemia or post-polycythemia vera myelofibrosis (MF) in close cooperation with the underlying molecular drivers. This inflammatory state is induced by a dynamic spectrum of inflammatory cytokines, although recent evidence points to the participation of additional soluble inflammatory mediators. Damage-associated molecular patterns (DAMPs) represent endogenous signals released upon cell death or damage which trigger a potent innate immune response. We assessed the contribution of two prototypical DAMPs, HMGB1 and S100A8/A9, to MF inflammation. Circulating HMGB1 and S100A8/A9 were elevated in MF patients in parallel to the degree of systemic inflammation and levels increased progressively during advanced disease stages. Patients with elevated DAMPs had higher frequency of adverse clinical features, such as anemia, and inferior survival, suggesting their contribution to disease progression. Monocytes, which are key players in MF inflammation, were identified as a source of S100A8/A9 but not HMGB1 release, while both DAMPs correlated with cell death parameters, such as serum LDH and cell-free DNA, indicating that passive release is an additional mechanism leading to increased DAMPs. HMGB1 and S100A8/A9 promote inflammation through binding to Toll-like receptor (TLR) 4, whereas the former also binds TLR2. Monocytes from MF patients were shown to be hyperactivated at baseline, as reflected by higher CD11b and tissue factor exposure and increased expression levels of proinflammatory cytokines IL-1β and IL-6. Patient monocytes showed preserved TLR4 and TLR2 expression and were able to mount normal or even exacerbated functional responses and cytokine upregulation following stimulation of TLR4 and TLR2. Elevated levels of endogenous TLR ligands HMGB1 and S100A8/A9 coupled to the finding of preserved or hyperreactive TLR-triggered responses indicate that DAMPs may promote monocyte activation and cytokine production in MF, fueling inflammation. Plasma IL-1β and IL-6 were elevated in MF and correlated with DAMPs levels, raising the possibility that DAMPs could contribute to cytokine generation in vivo. In conclusion, this study highlights that, in cooperation with classic proinflammatory cytokines, DAMPs represent additional inflammatory mediators that may participate in the generation of MF inflammatory state, potentially providing novel biomarkers of disease progression and new therapeutic targets.
Keywords: HMGB1; S100A8/A9; Toll-like receptors; inflammation; monocyte; myelofibrosis.
Copyright © 2024 De Luca, Goette, Lev, Baroni Pietto, Marin Oyarzún, Castro Ríos, Moiraghi, Sackmann, Kamiya, Verri, Caula, Fernandez, Vicente, Pose Cabarcos, Caruso, Camacho, Larripa, Khoury, Marta, Glembotsky and Heller.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
The alarmins high mobility group box protein 1 and S100A8/A9 display different inflammatory profiles after acute knee injury.Osteoarthritis Cartilage. 2022 Sep;30(9):1198-1209. doi: 10.1016/j.joca.2022.06.009. Epub 2022 Jul 6. Osteoarthritis Cartilage. 2022. PMID: 35809846
-
The roles of toll-like receptor 4, CD33, CD68, CD69, or CD147/EMMPRIN for monocyte activation by the DAMP S100A8/S100A9.Front Immunol. 2023 Mar 28;14:1110185. doi: 10.3389/fimmu.2023.1110185. eCollection 2023. Front Immunol. 2023. PMID: 37056775 Free PMC article.
-
High cell-free DNA is associated with disease progression, inflammasome activation and elevated levels of inflammasome-related cytokine IL-18 in patients with myelofibrosis.Front Immunol. 2023 Nov 16;14:1161832. doi: 10.3389/fimmu.2023.1161832. eCollection 2023. Front Immunol. 2023. PMID: 38035089 Free PMC article.
-
S100A8 and S100A9: DAMPs at the crossroads between innate immunity, traditional risk factors, and cardiovascular disease.Mediators Inflamm. 2013;2013:828354. doi: 10.1155/2013/828354. Epub 2013 Dec 22. Mediators Inflamm. 2013. PMID: 24453429 Free PMC article. Review.
-
S100A8/A9: From basic science to clinical application.Pharmacol Ther. 2016 Nov;167:120-131. doi: 10.1016/j.pharmthera.2016.07.015. Epub 2016 Aug 1. Pharmacol Ther. 2016. PMID: 27492899 Review.
Cited by
-
Inflammation-Related Factors S100A9 and TLR2 in Cardiomyocyte Hypertrophy.Curr Med Sci. 2025 Aug;45(4):819-830. doi: 10.1007/s11596-025-00096-2. Epub 2025 Jul 22. Curr Med Sci. 2025. PMID: 40694254
-
Low-Molecular-Weight Fucoidan Inhibits Thromboinflammation and Ameliorates Deep Vein Thrombosis via Targeting S100A8/A9.Mar Drugs. 2025 Apr 22;23(5):180. doi: 10.3390/md23050180. Mar Drugs. 2025. PMID: 40422770 Free PMC article.
-
Targeting Calprotectin S100A8/A9 to Overcome AML Progression in DNMT3A-Mutant Cells.Curr Med Sci. 2025 Jun;45(3):458-468. doi: 10.1007/s11596-025-00042-2. Epub 2025 Apr 23. Curr Med Sci. 2025. PMID: 40266434
-
Involvement of role of HMGB1-NLRP3 pathway in systemic disorders.Front Cell Dev Biol. 2025 Jul 4;13:1600596. doi: 10.3389/fcell.2025.1600596. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40688353 Free PMC article. Review.
References
-
- Tefferi A, Vaidya R, Caramazza D, Finke C, Lasho T, Pardanani A. Circulating interleukin (IL)-8, IL-2R, IL-12, and IL-15 levels are independently prognostic in primary myelofibrosis: a comprehensive cytokine profiling study. J Clin Oncol. (2011) 29:1356–63. doi: 10.1200/JCO.2010.32.9490 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

