A multilayer network analysis of Alzheimer's disease pathogenesis: Roles for p-tau, synaptic peptides, and physical activity
- PMID: 39394857
- PMCID: PMC11567865
- DOI: 10.1002/alz.14286
A multilayer network analysis of Alzheimer's disease pathogenesis: Roles for p-tau, synaptic peptides, and physical activity
Abstract
Introduction: In the aging brain, cognitive abilities emerge from the coordination of complex pathways arising from a balance between protective lifestyle and environmental factors and accumulation of neuropathologies.
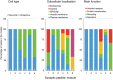
Methods: As part of the Rush Memory and Aging Project (n = 440), we measured accelerometer-based actigraphy, cognitive performance, and after brain autopsy, selected reaction monitoring mass spectrometry. Multilevel network analysis was used to examine the relationships among the molecular machinery of vesicular neurotransmission, Alzheimer's disease (AD) neuropathology, cognition, and late-life physical activity.
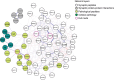
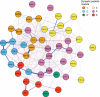
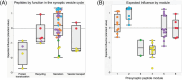
Results: Synaptic peptides involved in neuronal secretory function were the most influential contributors to the multilayer network, reflecting the complex interdependencies among AD pathology, synaptic processes, and late-life cognition. Older adults with lower physical activity evidenced stronger adverse relationships among phosphorylated tau peptides, markers of synaptic integrity, and tangle pathology.
Discussion: Network-based approaches simultaneously model interdependent biological processes and advance understanding of the role of physical activity in age-associated cognitive impairment.
Highlights: Network-based approaches simultaneously model interdependent biological processes. Secretory synaptic peptides were influential contributors to the multilayer network. Older adults with lower physical activity had adverse relationships among pathology. There was interdependence among phosphorylated tau, synaptic integrity, and tangles. Network methods elucidate the role of physical activity in cognitive impairment.
Keywords: Alzheimer's disease; aging; physical activity; post mortem brain; presynaptic proteins; proteomics; synaptopathy.
© 2024 The Author(s). Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
William G. Honer has received consulting fees or sat on paid advisory boards for: Translational Life Sciences, AbbVie, Boehringer Ingelheim, and Newron. The organizations cited above had no role in (and therefore did not influence) the design of the present study, the interpretation of results, and/or preparation of the manuscript. Andrea A. Jones, Alfredo Ramos‐Miguel, Kristina M. Gicas, Vladislav A. Petyuk, Sue E. Leurgans, Philip L. De Jager, Julie A. Schneider, David A. Bennett, and Kaitlin B. Casaletto have no potential conflict of interests. Author disclosures are available in the supporting information.
Figures


References
-
- McClelland JL. Emergence in cognitive science. Top Cogn Sci. 2010;2:751‐770. - PubMed
-
- Morris JC, Price JL. Pathologic correlates of nondemented aging, mild cognitive impairment, and early‐stage Alzheimer's disease. J Mol Neurosci. 2001;17:101‐118. - PubMed
-
- Hardy JA, Higgins GA. Alzheimer's disease: the amyloid cascade hypothesis. Science. 1992;256:184‐185. - PubMed
MeSH terms
Substances
Grants and funding
- U01 AG046152/AG/NIA NIH HHS/United States
- BIO22/ALZ/002/EiTB-Maratoia/BIOEF/Basque Government
- RYC-2016-19282/Alfredo Ramos-Miguel is a 'Ramón y Cajal' Researcher
- U01 AG061356/AG/NIA NIH HHS/United States
- MCIU-EHU/UPV/Alfredo Ramos-Miguel is a 'Ramón y Cajal' Researcher
- K23AG058752/AG/NIA NIH HHS/United States
- R01 AG017917/AG/NIA NIH HHS/United States
- R01 AG072475/AG/NIA NIH HHS/United States
- R01AG42210/AG/NIA NIH HHS/United States
- FRN-190708/CAPMC/ CIHR/Canada
- R01AG072475/AG/NIA NIH HHS/United States
- R01 AG042210/AG/NIA NIH HHS/United States
- U01AG46152/AG/NIA NIH HHS/United States
- BIO22/ALZ/002/Berrikuntza + Ikerketa + Osasuna Eusko Fundazioa
- K23 AG058752/AG/NIA NIH HHS/United States
- R01AG17917/AG/NIA NIH HHS/United States
- U01AG61356/AG/NIA NIH HHS/United States
- MOP-14037/CAPMC/ CIHR/Canada
LinkOut - more resources
Full Text Sources
Medical
Research Materials

