Soluble mesothelin-related peptide as a prognosticator in pleural mesothelioma patients receiving checkpoint immunotherapy
- PMID: 39395787
- PMCID: PMC11949723
- DOI: 10.1016/j.jtcvs.2024.10.005
Soluble mesothelin-related peptide as a prognosticator in pleural mesothelioma patients receiving checkpoint immunotherapy
Abstract
Background: Immune checkpoint therapy (ICT) has significantly impacted the treatment of malignant pleural mesothelioma (MPM). Despite some promising results from combination therapies, nearly half of MPM patients do not benefit, underscoring the urgent need for reliable predictive biomarkers. This study assesses the prognostic value of serum soluble mesothelin-related peptide (SMRP) and PD-L1 levels in MPM patients receiving ICT.
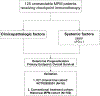
Methods: We conducted a retrospective analysis of 125 MPM patients treated with ICT by measuring pre-ICT serum levels of SMRP and PD-L1. We also examined the correlation of these serum levels with tumor mRNA expressions of mesothelin and PD-L1. Both univariable and multivariable Cox regression analyses were used to determine independent prognosticators for overall survival (OS). A prospective ICT clinical trial and our historical cohort were included for validation.
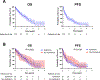
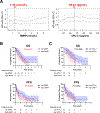
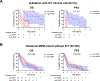
Results: Seventy-seven patients (62%) were treated with either anti-PD-(L)1 monotherapy, and the remaining 38% received combination ICT. Higher pre-ICT SMRP levels were observed in epithelioid MPM compared to nonepithelioid MPM. Serum PD-L1 levels did not differ significantly between the different histologic groups. Univariable analysis identified durable clinical benefit, development of immune-related adverse events, and SMRP levels as significantly associated with OS. Multivariable analysis confirmed SMRP as an independent prognostic factor, with lower levels (≤1.35 nmol/L) correlating with improved OS. The association of high SMRP with worse prognosis was validated in the prospective ICT clinical trial cohort and not in our historical cohort treated without ICT.
Conclusions: SMRP is a promising serum biomarker for predicting survival in MPM patients treated with ICT and warrants prospective investigation.
Keywords: SMRP; checkpoint immunotherapy; liquid biomarker; mesothelioma; prognostic biomarker; soluble PD-L1.
Copyright © 2024 The American Association for Thoracic Surgery. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of Interest Statement H.S.L. reports research funding from the National Institutes of Health, the Department of Defense, the Cancer Prevention Research Institute of Texas, the Helis Medical Research Foundation, and Dan L. Duncan Comprehensive Cancer Center, and investigator-initiated preclinical and clinical research funding from Samyang Biopharm and Momotaro-Gene. B.M.B. reports research funding from the National Institute of Health and Cancer Prevention Research Institute of Texas; clinical trial funding from AstraZeneca, Novartis, and Momotaro-Gene; and serving as a consultant in non–small cell lung cancer for AstraZeneca. R.T.R. reports research support from the National Institutes of Health, the American Association of Thoracic Surgery, and the DeGregorio Family Foundation; serving on the board of directors of the Mesothelioma Applied Research Foundation (nonremunerated); and providing expert legal opinion and serving on a speakers bureau for Merck. S.H.B has received IIT funding from Natera, Steris, and Medtronic; codeveloped instruments with Scanlan and holds other patents with Mayo Clinic; and has served as a speaker for Medtronic, AstraZeneca, and Medela. S.S.G. reports proctor and speaker honoraria from Intuitive Surgical. All other authors reported no conflicts of interest. The Journal policy requires editors and reviewers to disclose conflicts of interest and to decline handling or reviewing manuscripts for which they may have a conflict of interest. The editors and reviewers of this article have no conflicts of interest.
Figures


References
-
- Bueno R, Stawiski EW, Goldstein LD, et al. Comprehensive genomic analysis of malignant pleural mesothelioma identifies recurrent mutations, gene fusions and splicing alterations. Nat Genet. 2016;48:407–416. - PubMed
-
- NCCN Guidelines Mesothelioma: Pleural (Version: 1.2024). 2024, at https://www.nccn.org/login?ReturnURL=https://www.nccn.org/professionals/....)
-
- Cancer Facts & Figures 2023. 2023. (Accessed 10/03/2023, 2023, at cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/202....)
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

