How a paramyxovirus fusion/entry complex adapts to escape a neutralizing antibody
- PMID: 39396053
- PMCID: PMC11470942
- DOI: 10.1038/s41467-024-53082-y
How a paramyxovirus fusion/entry complex adapts to escape a neutralizing antibody
Abstract
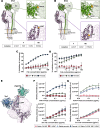
Paramyxoviruses including measles, Nipah, and parainfluenza viruses are public health threats with pandemic potential. Human parainfluenza virus type 3 (HPIV3) is a leading cause of illness in pediatric, older, and immunocompromised populations. There are no approved vaccines or therapeutics for HPIV3. Neutralizing monoclonal antibodies (mAbs) that target viral fusion are a potential strategy for mitigating paramyxovirus infection, however their utility may be curtailed by viral evolution that leads to resistance. Paramyxoviruses enter cells by fusing with the cell membrane in a process mediated by a complex consisting of a receptor binding protein (HN) and a fusion protein (F). Existing atomic resolution structures fail to reveal physiologically relevant interactions during viral entry. We present cryo-ET structures of pre-fusion HN-F complexes in situ on surfaces of virions that evolved resistance to an anti-HPIV3 F neutralizing mAb. Single mutations in F abolish mAb binding and neutralization. In these complexes, the HN protein that normally restrains F triggering has shifted to uncap the F apex. These complexes are more readily triggered to fuse. These structures shed light on the adaptability of the pre-fusion HN-F complex and mechanisms of paramyxoviral resistance to mAbs, and help define potential barriers to resistance for the design of mAbs.
© 2024. The Author(s).
Conflict of interest statement
T.C.M., M.P., and A.M. are listed as inventors on a provisional patent related to this work, “Subnanometer Structure of an Enveloped Virus Fusion Complex on Viral Surface Reveals New Entry Mechanisms.” The other authors do not have competing interests.
Figures


References
Publication types
MeSH terms
Substances
Associated data
- Dryad/10.5061/dryad.g1jwstqz6
- BioProject/PRJNA1083633
- Actions
- Actions
Grants and funding
- R01 AI160953/AI/NIAID NIH HHS/United States
- R01 AI114736/AI/NIAID NIH HHS/United States
- U19 AI181984/AI/NIAID NIH HHS/United States
- AI121349/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (Division of Intramural Research of the NIAID)
- AI160961/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (Division of Intramural Research of the NIAID)
- AI160953/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (Division of Intramural Research of the NIAID)
- AI152275/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (Division of Intramural Research of the NIAID)
- AI114736/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (Division of Intramural Research of the NIAID)
- R01 AI160961/AI/NIAID NIH HHS/United States
- R01 AI175362/AI/NIAID NIH HHS/United States
- F32 AI152275/AI/NIAID NIH HHS/United States
- R01 AI121349/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

