The New Era of Total Neoadjuvant FLOT Therapy for Locally Advanced, Resectable Gastric Cancer: A Propensity-Matched Comparison With Standard Perioperative Therapy
- PMID: 39400342
- PMCID: PMC12044282
- DOI: 10.1002/jso.27934
The New Era of Total Neoadjuvant FLOT Therapy for Locally Advanced, Resectable Gastric Cancer: A Propensity-Matched Comparison With Standard Perioperative Therapy
Abstract
Background: The FLOT 4-AIO trial established the docetaxel-based regimen's superiority over epirubicin-based triplet therapy in terms of survival rates and acceptable toxicity for locally advanced resectable gastric (LARGC). Yet, fewer than half of the patients achieved completion of eight prescribed FLOT cycles. We proposed that administering all FLOT cycles in the form of total neoadjuvant therapy may improve completion rates and downstaging. This study contrasted total neoadjuvant therapy (FLOT x8) with standard neoadjuvant therapy (FLOT 4+4) for patients LARGC adenocarcinoma who underwent curative resection with routine D2 lymphadenectomy, focusing on histopathological outcomes, toxicity, and survival outcomes.
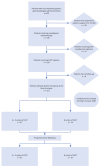
Methods: We reviewed patients with histologically confirmed advanced clinical stage cT2 or higher, nodal positive stage (cN+), or both, with resectable gastric tumors and no distant metastases (January 2017 to July 2023). We divided patients into two groups, FLOT 4+4 and FLOT x8; FLOT 4+4 patients underwent four preoperative and four postoperative bi-weekly cycles of docetaxel, oxaliplatin, leucovorin, and fluorouracil, while FLOT x8 patients received all eight cycles preoperatively after a gradual practice change starting from January 2020. Propensity score matching adjusted for age, clinical stage, tumor location, and histology.
Results: Of the 77 patients in the FLOT x8 group, 37 were propensity-matched to an equal number in the FLOT 4+4 group. Demographics, duration of surgery, and hospital stay showed no significant differences between the groups. The FLOT x8 group exhibited a significantly higher all-cycle completion rate at 89.1% compared to FLOT 4+4's 67.6% (p < 0.01). Both groups demonstrated comparable hematological and non-hematological toxicity rates, Clavien-Dindo ≥ 3 complications, and CAP tumor regression grades. The mean number of harvested lymph nodes was 42.5 and 41.2 in the FLOT 4+4 and FLOT x8 groups, respectively. Similar rates of disease-free survival and overall survival were noted in both groups, despite a trend toward a higher pathological complete response rate, albeit not statistically significant (8.1% vs. 18.9%, p = 0.29), in the FLOT x8 group at a median follow-up of 36 months.
Conclusion: Total neoadjuvant therapy with the FLOT x8 protocol corresponds to higher treatment completion rates, a safety profile similar to standard perioperative therapy, and a twofold increase in complete pathological response. Further research on long-term oncological outcomes is needed to confirm the effectiveness of total neoadjuvant therapy.
Keywords: FLOT therapy; gastric cancer; perioperative therapy; total neoadjuvant therapy.
© 2024 The Author(s). Journal of Surgical Oncology published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Reim D., Loos M., Vogl F., et al., “Prognostic Implications of the Seventh Edition of the International Union Against Cancer Classification for Patients With Gastric Cancer: The Western Experience of Patients Treated in a Single‐Center European Institution,” Journal of Clinical Oncology 31, no. 2 (2013): 263–271. - PubMed
-
- Cunningham D., Allum W. H., Stenning S. P., et al., “Perioperative Chemotherapy Versus Surgery Alone for Resectable Gastroesophageal Cancer,” New England Journal of Medicine 355, no. 1 (2006): 11–20. - PubMed
-
- Al‐Batran S. E., Homann N., Pauligk C., et al., “Perioperative Chemotherapy With Fluorouracil Plus Leucovorin, Oxaliplatin, and Docetaxel Versus Fluorouracil or Capecitabine Plus Cisplatin and Epirubicin for Locally Advanced, Resectable Gastric or Gastro‐Oesophageal Junction Adenocarcinoma (FLOT 4): A Randomised, Phase 2/3 Trial,” Lancet 393, no. 10184 (2019): 1948–1957. - PubMed
-
- Ajani J. A., D'Amico T. A., Bentrem D. J., et al., “Esophageal and Esophagogastric Junction Cancers, Version 2.2023, NCCN Clinical Practice Guidelines in Oncology,” Journal of the National Comprehensive Cancer Network 21, no. 4 (2023): 393–422. - PubMed
-
- Brierley J. D., Gospodarowicz M. K., and Wittekind C., TNM Classification of Malignant Tumours (Hoboken, NJ, USA: John Wiley & Sons, 2017).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

