Nanoplatelets modified with RVG for targeted delivery of miR-375 and temozolomide to enhance gliomas therapy
- PMID: 39402578
- PMCID: PMC11476726
- DOI: 10.1186/s12951-024-02895-6
Nanoplatelets modified with RVG for targeted delivery of miR-375 and temozolomide to enhance gliomas therapy
Abstract
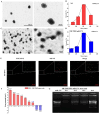
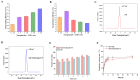
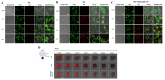
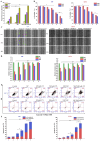
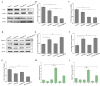
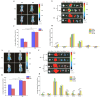
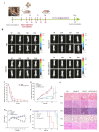
Gliomas are one of the most frequent primary brain tumors and pose a serious threat to people's lives and health. Platelets, a crucial component of blood, have been applied as drug delivery carriers for disease diagnosis and treatment. In this study, we designed engineered nanoplatelets for targeted delivery of therapeutic miR-375 and temozolomide (TMZ, a first-line glioma treatment agent) to enhance glioma therapy. Nanoplatelets were prepared through mild ultrasound, TMZ and miR-375 were co-loaded through ultrasound and electrostatic interactions, respectively, to combine chemotherapy with gene therapy against glioma. To improve the blood brain barrier (BBB) crossing efficiency and glioma targeting ability, the nanoplatelets were modified with central nervous system-specific rabies viral glycoprotein peptide (RVG) through thiol-maleimide click reaction. The RVG modified nanoplatelets co-loaded TMZ and miR-375 (NR/TMZ/miR-375) not only inherited the good stability and remarkable biocompatibility of platelets, but also promoted the cellular uptake and penetration of glioma tissues, and effectively induced cell apoptosis to enhance the therapeutic effect of drugs. In vivo studies showed that NR/TMZ/miR-375 significantly increased the circulation time of TMZ, and exhibited superior combined antitumor effects. In summary, this multifunctional 'natural' nanodrug delivery system provides a potent, scalable, and safety approach for platelet-based combined cancer chemotherapy and gene therapy.
Keywords: Gliomas; MiR-375; Nanoplatelets; TMZ; Targeted drug delivery.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- Sousa F. Emerging cytokine delivery with nanomedicine for brain cancer treatment. Expert Opin Drug Deliv. 2024;21:513–6. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

