Transition-metal-free synthesis of arylboronates via thermal generation of aryl radicals from triarylbismuthines in air
- PMID: 39403306
- PMCID: PMC11472659
- DOI: 10.3762/bjoc.20.216
Transition-metal-free synthesis of arylboronates via thermal generation of aryl radicals from triarylbismuthines in air
Abstract
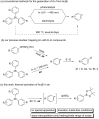
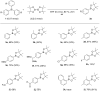
A simple and versatile synthesis of arylboronates has been achieved by using triarylbismuthines as aryl radical sources under transition-metal-free and open-air conditions. Conventional methods required photoirradiation or electrolysis to generate aryl radicals from triarylbismuthines. In this study, it was found that simply heating the solution of triarylbismuthines in benzotrifluoride (BTF) in air successfully led to the generation of aryl radicals, and the subsequent reaction with bis(pinacolato)diboron afforded a variety of arylboronates in moderate to good yields.
Keywords: arylboronates; bis(pinacolato)diboron; radical reactions; transition-metal-free synthesis; triarylbismuthines.
Copyright © 2024, Yamamoto et al.
Figures




References
LinkOut - more resources
Full Text Sources
Miscellaneous
