Make it STING: nanotechnological approaches for activating cGAS/STING as an immunomodulatory node in osteosarcoma
- PMID: 39403376
- PMCID: PMC11471590
- DOI: 10.3389/fimmu.2024.1403538
Make it STING: nanotechnological approaches for activating cGAS/STING as an immunomodulatory node in osteosarcoma
Abstract
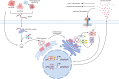
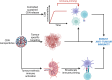
Osteosarcoma is a highly aggressive bone cancer primarily affecting children, adolescents, and young adults. The current gold standard for treatment of osteosarcoma patients consists of two to three rounds of chemotherapy, followed by extensive surgical intervention from total limb reconstruction to amputation, followed by additional rounds of chemotherapy. Although chemotherapy has advanced the treatment of osteosarcoma significantly, the overall 5-year survival rate in resistant forms of osteosarcoma is still below 20%. The interaction between cancer and the immune system has long been recognized as a critical aspect of tumour growth. Tumour cells within the tumour microenvironment (TME) suppress antitumour immunity, and immunosuppressive cells and cytokines provide the extrinsic factors of tumour drug resistance. Emerging research demonstrates an immunostimulatory role for the cGAS/STING pathway in osteosarcoma, typically considered an immune-cold or immunosuppressed cancer type. cGAS/STING signalling appears to drive an innate immune response against tumours and potentiates the efficacy of other common therapies including chemo and radiotherapy. Nanotechnological delivery systems for improved therapy delivery for osteosarcoma have also been under investigation in recent years. This review provides an overview of cGAS/STING signalling, its divergent roles in the context of cancer, and collates current research which activates cGAS/STING as an adjuvant immunomodulatory target for the treatment of osteosarcoma. It will also discuss current nanotechnological delivery approaches that have been developed to stimulate cGAS/STING. Finally, it will highlight the future directions that we believe will be central to the development of this transformative field.
Keywords: STING; cGAS; drug delivery; immunotherapy; innate immunity; nanotechnology; osteosarcoma.
Copyright © 2024 O’Donoghue and Freeman.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Sirichativapee W, Wisanuyotin T, Pattanittum P, Paholpak P, Laupattarakasem P, Srisodaphol W, et al. Chemotherapy for treating high-grade osteosarcoma in children and young adults. Cochrane Database Syst Rev. (2018) 2018:CD012372. doi: 10.1002/14651858.CD012372.pub2 - DOI
-
- Freeman FE, Dosta P, Shanley LC, Ramirez Tamez N, Riojas Javelly CJ, Mahon OR, et al. Localized Nanoparticle-Mediated Delivery of miR-29b Normalizes the Dysregulation of Bone Homeostasis Caused by Osteosarcoma whilst Simultaneously Inhibiting Tumor Growth. Adv Mater. (2023) 35:2207877. doi: 10.1002/adma.202207877 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

