Unlocking the secrets of NPSLE: the role of dendritic cell-secreted CCL2 in blood-brain barrier disruption
- PMID: 39403387
- PMCID: PMC11472714
- DOI: 10.3389/fimmu.2024.1343805
Unlocking the secrets of NPSLE: the role of dendritic cell-secreted CCL2 in blood-brain barrier disruption
Abstract
Background: This study employed RNA-seq technology and meta-analysis to unveil the molecular mechanisms of neuropsychiatric systemic lupus erythematosus (NPSLE) within the central nervous system.
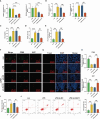
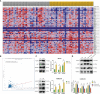
Methods: Downloaded transcriptomic data on systemic lupus erythematosus (SLE) from the Gene Expression Omnibus (GEO) and analyzed differential genes in peripheral blood samples of NPSLE patients and healthy individuals. Employed WGCNA to identify key genes related to cognitive impairment and validated findings via RNA-seq. Conducted GO, KEGG, and GSEA analyses, and integrated PPI networks to explore gene regulatory mechanisms. Assessed gene impacts on dendritic cells and blood-brain barrier using RT-qPCR, ELISA, and in vitro models.
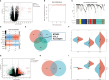
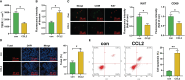
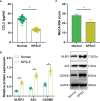
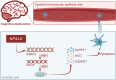
Results: Public databases and RNA-seq data have revealed a significant upregulation of CCL2 (C-C motif chemokine ligand 2) in the peripheral blood of both SLE and NPSLE patients, primarily secreted by mature dendritic cells. Furthermore, the secretion of CCL2 by mature dendritic cells may act through the RSAD2-ISG15 axis and is associated with the activation of the NLRs (Nod Like Receptor Signaling Pathway) signaling pathway in vascular endothelial cells. Subsequent in vitro cell experiments confirmed the high expression of CCL2 in peripheral blood dendritic cells of NPSLE patients, with its secretion being regulated by the RSAD2-ISG15 axis and inducing vascular endothelial cell pyroptosis through the activation of the NLRs signaling pathway. Clinical trial results ultimately confirmed that NPSLE patients exhibiting elevated CCL2 expression also experienced cognitive decline.
Conclusions: The secretion of CCL2 by dendritic cells induces pyroptosis in vascular endothelial cells, thereby promoting blood-brain barrier damage and triggering cognitive impairment in patients with systemic lupus erythematosus.
Keywords: CCL2; NLR signaling pathway; RNA-seq; dendritic cells; neuropsychiatric systemic lupus erythematosus.
Copyright © 2024 Wang, Zheng, Wang and Jia.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Blood-brain barrier disruption and neuroinflammation as pathophysiological mechanisms of the diffuse manifestations of neuropsychiatric systemic lupus erythematosus.Autoimmun Rev. 2019 Apr;18(4):426-432. doi: 10.1016/j.autrev.2018.12.004. Epub 2019 Feb 11. Autoimmun Rev. 2019. PMID: 30763633 Review.
-
Differential activation mechanisms of serum C5a in lupus nephritis and neuropsychiatric systemic lupus erythematosus.Mod Rheumatol. 2017 Mar;27(2):292-297. doi: 10.1080/14397595.2016.1193965. Epub 2016 Jun 20. Mod Rheumatol. 2017. PMID: 27319578
-
Upregulation of microRNA-762 suppresses the expression of GIPC3 in systemic lupus erythematosus and neuropsychiatric systemic lupus erythematosus.Immun Inflamm Dis. 2022 Nov;10(11):e719. doi: 10.1002/iid3.719. Immun Inflamm Dis. 2022. PMID: 36301034 Free PMC article.
-
Blood-brain barrier damages and intrathecal synthesis of anti-N-methyl-D-aspartate receptor NR2 antibodies in diffuse psychiatric/neuropsychological syndromes in systemic lupus erythematosus.Arthritis Res Ther. 2014 Mar 21;16(2):R77. doi: 10.1186/ar4518. Arthritis Res Ther. 2014. PMID: 24655341 Free PMC article.
-
Insights into the role of neutrophils in neuropsychiatric systemic lupus erythematosus: Current understanding and future directions.Front Immunol. 2022 Aug 9;13:957303. doi: 10.3389/fimmu.2022.957303. eCollection 2022. Front Immunol. 2022. PMID: 36016935 Free PMC article. Review.
Cited by
-
Relationship Between Cognitive Impairment and Quality of Life in Systemic Lupus Erythematosus: A Multimodal Neuroimaging Perspective.Int J Gen Med. 2025 Aug 7;18:4327-4336. doi: 10.2147/IJGM.S533375. eCollection 2025. Int J Gen Med. 2025. PMID: 40792067 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

