Quantitative analysis of islet prohormone convertase 1/3 expression in human pancreas donors with diabetes
- PMID: 39404844
- PMCID: PMC11604696
- DOI: 10.1007/s00125-024-06275-5
Quantitative analysis of islet prohormone convertase 1/3 expression in human pancreas donors with diabetes
Abstract
Aims/hypothesis: Islet prohormone-processing enzymes convert peptide hormone precursors to mature hormones. Defective beta cell prohormone processing and the release of incompletely processed peptide hormones are observed prior to the onset of diabetes, yet molecular mechanisms underlying impaired prohormone processing during the development of diabetes remains largely unknown. Previous studies have shown that prohormone convertase 1/3 (PC1/3) protein and mRNA expression levels are reduced in whole islets from donors with type 1 diabetes, although whether PC1/3-mediated prohormone processing in alpha and beta cells is disrupted in type 1 diabetes remained to be explored. Herein, we aimed to analyse the expression of PC1/3 in islets from non-diabetic donors, autoantibody-positive donors and donors diagnosed with type 1 diabetes or type 2 diabetes.
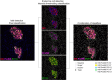
Methods: Immunostaining and high-dimensional image analysis were performed on pancreatic sections from a cross-sectional cohort of 54 donors obtained from the Network for Pancreatic Organ Donors with Diabetes (nPOD) repository, to evaluate PC1/3 expression patterns in islet alpha, beta and delta cells at different stages of diabetes.
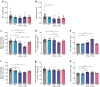
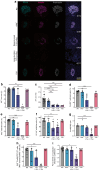
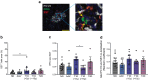
Results: Alpha and beta cell morphology were altered in donors with type 1 diabetes, including decreased alpha and beta cell size. As expected, the insulin-positive and PC1/3-positive areas in the islets were both reduced, and this was accompanied by a reduced percentage of PC1/3-positive and insulin-positive/PC1/3-positive cells in islets. PC1/3 and insulin co-localisation was also reduced. The glucagon-positive area, as well as the percentage of glucagon-positive and glucagon-positive/PC1/3-positive cells in islets, was increased. PC1/3 and glucagon co-localisation was also increased in donors with type 1 diabetes. The somatostatin-positive cell area and somatostatin staining intensity were elevated in islets from donors with recent-onset type 1 diabetes.
Conclusions/interpretation: Our high-resolution histomorphological analysis of human pancreatic islets from donors with and without diabetes has uncovered details of the cellular origin of islet prohormone peptide processing defects. Reduced beta cell PC1/3 and increased alpha cell PC1/3 in islets from donors with type 1 diabetes pinpointed the functional deterioration of beta cells and the concomitant potential increase in PC1/3 usage for prohormone processing in alpha cells during the pathogenesis of type 1 diabetes. Our finding of PC1/3 loss in beta cells may inform the discovery of new prohormone biomarkers as indicators of beta cell dysfunction, and the finding of elevated PC1/3 expression in alpha cells may encourage the design of therapeutic targets via leveraging alpha cell adaptation in diabetes.
Keywords: Beta cell dysfunction; Prohormone convertase 1/3; Proinsulin; Quantitative image analysis.
© 2024. The Author(s).
Conflict of interest statement
Acknowledgements: We thank the organ donors and their families. We thank the Network for Pancreatic Organ Donors (nPOD) and especially thank I. Kusmartseva and M. Yang (Department of Pathology, Immunology, and Laboratory Medicine, University of Florida, USA) for pancreatic tissue sample procurement and distribution. We also thank P.-I. Petropoulou (Institute of Diabetes Research, Helmholtz Zentrum München, Germany) for helpful suggestions to the project. We acknowledge technical support from the Imaging Core at the Canucks for Kids Childhood Diabetes Laboratory at BC Children’s Hospital Research Institute. Data availability: Raw data and images are available upon request. Funding: Open Access funding enabled and organized by Projekt DEAL. This work was supported by grants from Breakthrough T1D (3-COE-2022-1103-M-B) through Breakthrough T1D Centre of Excellence at University of British Columbia and Canadian Institutes of Health and Research (CIHR PJT-153156) to CBV, a Breakthrough T1D Advanced Postdoctoral Fellowship (3-APF-2022–1141-A-N) to Y-CC, a University of British Columbia Faculty of Medicine Summer Studentship (to KG), and a career development award from Breakthrough T1D (5-CDA-2020–949-A-N) to TR-C. Research in the Rodriguez-Calvo laboratory is supported by IMI2-JU under grant agreement no. 115797 (INNODIA) and no. 945268 (INNODIA HARVEST). This Joint Undertaking receives support from the Union’s Horizon 2020 research and innovation programme and EFPIA, Breakthrough T1D and The Leona M. and Harry B. Helmsley Charitable Trust. The study sponsor/funder was not involved in the design of the study; the collection, analysis and interpretation of data; writing the report; and did not impose any restrictions regarding the publication of the report. Authors’ relationships and activities: The authors declare that there are no relationships or activities that might bias, or be perceived to bias, their work. Contribution statement: PSA contributed to study design, data analysis, interpretation of data and drafted the manuscript. Y-CC contributed to the study conception and design, acquisition, analysis and interpretation of data and drafted the manuscript. YL and SB contributed to data analysis, and reviewed the manuscript. KG contributed to data acquisition and reviewed the manuscript. CBV contributed to the conception and design, interpretation of data and reviewed the manuscript. TR-C contributed to interpretation of data, and edited and reviewed the manuscript. PSA and Y-CC contributed equally to this work. All authors have approved the final version of the manuscript to be published. Both CBV and TR-C are guarantors of this study.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

