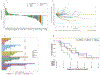Histology-Specific Clinical Trial of Lenvatinib and Pembrolizumab in Patients with Sarcoma
- PMID: 39405335
- PMCID: PMC11730159
- DOI: 10.1158/1078-0432.CCR-24-2519
Histology-Specific Clinical Trial of Lenvatinib and Pembrolizumab in Patients with Sarcoma
Abstract
Purpose: Survival of patients with metastatic sarcoma remains poor, and there is a pressing need for new therapies. Most sarcoma subtypes are not responsive to immune checkpoint inhibition alone. Lenvatinib, a multireceptor tyrosine kinase inhibitor targeting tumor vasculature, has an immunomodulatory activity that contributes to its antitumor effects. Therefore, we hypothesized that a combination of lenvatinib and pembrolizumab would lead to improved clinical outcomes in patients with sarcoma.
Patients and methods: This was an open-label, single-arm study of lenvatinib and pembrolizumab in the following cohorts: (A) leiomyosarcoma, (B) undifferentiated pleomorphic sarcoma (UPS), (C) vascular sarcomas (angiosarcoma and epithelioid hemangioendothelioma), (D) synovial sarcoma or malignant peripheral nerve sheath tumor (MPNST), and (E) bone sarcomas (osteosarcoma and chondrosarcoma). The primary endpoint was the best overall response (BOR) rate documented by RECIST v1.1 by 27 weeks in each cohort, with a threshold of ≥2 responses among 10 patients. Secondary endpoints included progression-free survival, overall survival, duration of response, and safety.
Results: Forty-six patients were evaluable for the primary endpoint, which was met in the UPS and MPNST/synovial cohorts (BOR rates by 27 weeks of 25% and 30%, respectively). There were seven partial responses overall with additional responses noted in angiosarcoma and osteosarcoma. Treatment-related adverse events of any grade and grade 3 or higher occurred in 50/51 (98%) and 29/51 (57%) of patients, respectively.
Conclusions: We observed durable responses in MPNST, synovial sarcoma, and osteosarcoma. Patients with UPS and angiosarcoma also responded. Further exploration of this approach is warranted to confirm activity and determine optimal dosing schedules.
©2024 American Association for Cancer Research.
Conflict of interest statement
Figures
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous