Retinol-binding protein type 1 expression predicts poor prognosis in head and neck squamous cell carcinoma
- PMID: 39407127
- PMCID: PMC11476480
- DOI: 10.1186/s12885-024-12565-3
Retinol-binding protein type 1 expression predicts poor prognosis in head and neck squamous cell carcinoma
Abstract
Background: Head and neck squamous cell carcinoma (HNSCC) is the sixth most prevalent malignancy worldwide, with high incidence and poor survival rates. RBP1 is highly expressed in several kinds of cancer and plays a potential prognostic factor. However, the relationship between RBP1 and HNSCC were analyzed based on The Cancer Genome Atlas (TCGA) database.
Materials and methods: RBP1 expression and clinical information were obtained from the Cancer Genome Atlas (TCGA) database. Tumor tissue and adjacent normal tissue of 6 HNSCC patients were collected to analyze the RBP1 mRNA expression level by quantitative PCR. Cox regression analysis was used to evaluate the prognostic values of RBP1 and clinical data in HNSCC. A nomogram was also established to predict the impact of RBP1 on prognosis based on Cox multivariate results. The methylation level of RBP1 in HNSC and its prognosis were analyzed in UALACN and MethSurv. Finally, the potential biological functions of RBP1 were investigated using gene set enrichment analysis (GSEA) and single sample GSEA (ssGSEA).
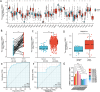
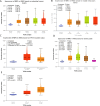
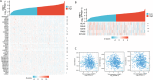
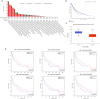
Results: The mRNA expression levels of RBP1 were highly expressed in HNSCC tissue. The Cox analyses demonstrate that highly-expressed RBP1 is an independent prognosis marker(P < 0.05). ROC curve analysis showed that performances of RBP1 (area under the ROC curve: 0.887, sensitivity: 84.1%, specificity: 79.9%). The methylation was increased in HNSCC patients compared with normal subjects(P < 0.05) and was associated with better prognosis at sites cg06208339, cg12298268, cg12497564, cg15288618, cg20532370, cg23448348. Additionally, RBP1 expression is mildly associated with immune cell infiltration and immunological checkpoints.
Conclusion: RBP1 is overexpressed and associated with poor patient prognosis in head and neck squamous cell carcinoma.
Keywords: Bioinformatic analysis; Head and neck squamous cell carcinoma; Prognosis biomarker; RBP1.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. - PubMed
-
- Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359–386. - PubMed
-
- Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM, Pineros M, Znaor A, Bray F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2019;144(8):1941–53. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

