Evaluation of Retinal Structure and Visual Function in Blue Cone Monochromacy to Develop Clinical Endpoints for L-opsin Gene Therapy
- PMID: 39408969
- PMCID: PMC11477341
- DOI: 10.3390/ijms251910639
Evaluation of Retinal Structure and Visual Function in Blue Cone Monochromacy to Develop Clinical Endpoints for L-opsin Gene Therapy
Abstract
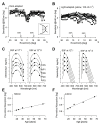
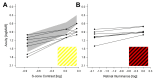
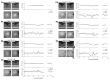
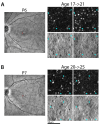
L-cone opsin expression by gene therapy is a promising treatment for blue cone monochromacy (BCM) caused by congenital lack of long- and middle-wavelength-sensitive (L/M) cone function. Eight patients with BCM and confirmed pathogenic variants at the OPN1LW/OPN1MW gene cluster participated. Optical coherence tomography (OCT), chromatic perimetry, chromatic microperimetry, chromatic visual acuity (VA), and chromaticity thresholds were performed with unmodified commercial equipment and/or methods available in the public domain. Adaptive optics scanning laser ophthalmoscope (AOSLO) imaging was performed in a subset of patients. Outer retinal changes were detectable by OCT with an age-related effect on the foveal disease stage. Rod and short-wavelength-sensitive (S) cone functions were relatively retained by perimetry, although likely impacted by age-related increases in the pre-retinal absorption of short-wavelength lights. The central macula showed a large loss of red sensitivity on dark-adapted microperimetry. Chromatic VAs with high-contrast red gratings on a blue background were not detectable. Color vision was severely deficient. AOSLO imaging showed reduced total cone density with majority of the population being non-waveguiding. This study developed and evaluated specialized outcomes that will be needed for the determination of efficacy and safety in human clinical trials. Dark-adapted microperimetry with a red stimulus sampling the central macula would be a key endpoint to evaluate the light sensitivity improvements. VA changes specific to L-opsin can be measured with red gratings on a bright blue background and should also be considered as outcome measures in future interventional trials.
Keywords: color vision; microperimetry; outcome measures; perimetry.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




References
-
- Wissinger B., Baumann B., Buena-Atienza E., Ravesh Z., Cideciyan A.V., Stingl K., Audo I., Meunier I., Bocquet B., Traboulsi E.I., et al. The landscape of submicroscopic structural variants at the OPN1LW/OPN1MW gene cluster on Xq28 underlying blue cone monochromacy. Proc. Natl. Acad. Sci. USA. 2022;119:e2115538119. doi: 10.1073/pnas.2115538119. - DOI - PMC - PubMed
-
- Cideciyan A.V., Hufnagel R.B., Carroll J., Sumaroka A., Luo X., Schwartz S.B., Dubra A., Land M., Michaelides M., Gardner J.C., et al. Human cone visual pigment deletions spare sufficient photoreceptors to warrant gene therapy. Hum. Gene Ther. 2013;24:993–1006. doi: 10.1089/hum.2013.153. - DOI - PMC - PubMed
-
- Luo X., Cideciyan A.V., Iannaccone A., Roman A.J., Ditta L.C., Jennings B.J., Yatsenko S.A., Sheplock R., Sumaroka A., Swider M., et al. Blue cone monochromacy: Visual function and efficacy outcome measures for clinical trials. PLoS ONE. 2015;10:e0125700. doi: 10.1371/journal.pone.0125700. - DOI - PMC - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

