Utilizing Adenovirus Knob Proteins as Carriers in Cancer Gene Therapy Amidst the Presence of Anti-Knob Antibodies
- PMID: 39409008
- PMCID: PMC11476472
- DOI: 10.3390/ijms251910679
Utilizing Adenovirus Knob Proteins as Carriers in Cancer Gene Therapy Amidst the Presence of Anti-Knob Antibodies
Abstract
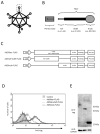
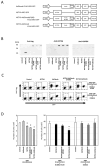
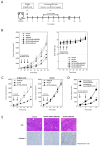
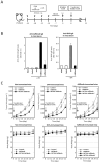
Numerous gene therapy drugs for cancer have received global approval, yet their efficacy against solid tumors remains inadequate. Our previous research indicated that the fiber protein, a component of the adenovirus capsid, can propagate from infected cells to neighboring cells that express the adenovirus receptor. We hypothesize that merging this fiber protein with an anti-cancer protein could enable the anti-cancer protein to disseminate around the transfected cells, presenting a novel approach to cancer gene therapy. In our study, we discovered that the knob region of the adenovirus type 5 fiber protein is the smallest unit capable of spreading to adjacent cells in a receptor-specific manner. We also showed that the recombinant knob protein infiltrates cells after dispersing to surrounding cells. To assess the potential of the knob protein to augment gene therapy for solid tumors in mice, we expressed a fusion gene of the A subunit of cytotoxic cholera toxin and the knob region in mouse tumors. We found that this fusion protein only inhibited tumor growth in receptor-expressing mouse melanomas, and this inhibitory effect persisted even in mice with anti-knob antibodies. Our study's findings propose a novel cancer gene therapy strategy that enhances therapeutic effects by specifically delivering therapeutic proteins, expressed from in vivo administered genes, to target molecules. This outcome offers a fresh perspective on gene therapy for solid cancers, and we anticipate that knob proteins will serve as a platform for this method.
Keywords: cancer; gene therapy; knob protein; neutralizing antibodies; targeting.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures


References
-
- Mast T.C., Kierstead L., Gupta S.B., Nikas A.A., Kallas E.G., Novitsky V., Mbewe B., Pitisuttithum P., Schechter M., Vardas E., et al. International Epidemiology of Human Pre-Existing Adenovirus (Ad) Type-5, Type-6, Type-26 and Type-36 Neutralizing Antibodies: Correlates of High Ad5 Titers and Implications for Potential HIV Vaccine Trials. Vaccine. 2010;28:950–957. doi: 10.1016/j.vaccine.2009.10.145. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

