Sodium Hydrosulfide Protects Rats from Hypobaric-Hypoxia-Induced Acute Lung Injury
- PMID: 39409062
- PMCID: PMC11477091
- DOI: 10.3390/ijms251910734
Sodium Hydrosulfide Protects Rats from Hypobaric-Hypoxia-Induced Acute Lung Injury
Abstract
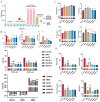
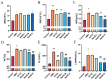
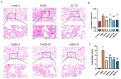
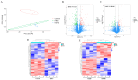
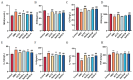
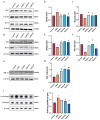
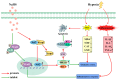
Hydrogen sulfide (H2S), as a key gas signaling molecule, plays an important role in regulating various diseases, with appropriate concentrations providing antioxidative, anti-inflammatory, and anti-apoptotic effects. The specific role of H2S in acute hypoxic injury remains to be clarified. This study focuses on the H2S donor sodium hydrosulfide (NaHS) and explores its protective effects and mechanisms against acute hypoxic lung injury. First, various mouse hypoxia models were established to evaluate H2S's protection in hypoxia tolerance. Next, a rat model of acute lung injury (ALI) induced by hypoxia at 6500 m above sea level for 72 h was created to assess H2S's protective effects and mechanisms. Evaluation metrics included blood gas analysis, blood routine indicators, lung water content, and lung tissue pathology. Additionally, LC-MS/MS and bioinformatic analyses were combined in performing quantitative proteomics on lung tissues from the normoxic control group, the hypoxia model group, and the hypoxia model group with NaHS treatment to preliminarily explore the protective mechanisms of H2S. Further, enzyme-linked immunosorbent assays (ELISA) were used to measure oxidative stress markers and inflammatory factors in rat lung tissues. Lastly, Western blot analysis was performed to detect Nrf2, HO-1, P-NF-κB, NF-κB, HIF-1α, Bcl-2, and Bax proteins in lung tissues. Results showed that H2S exhibited significant anti-hypoxic effects in various hypoxia models, effectively modulating blood gas and blood routine indicators in ALI rats, reducing pulmonary edema, improving lung tissue pathology, and alleviating oxidative stress, inflammatory responses, and apoptosis levels.
Keywords: acute lung injury; anti-apoptotic; anti-inflammatory; antioxidant; high-altitude hypoxia; hydrogen sulfide; proteomics.
Conflict of interest statement
The authors declare that they do not have any conflicts of interest related to the contents of this article.
Figures
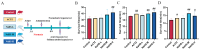


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

