Compound Heterozygous RYR1 Variants in a Patient with Severe Congenital Myopathy: Case Report and Comparison with Additional Cases of Recessive RYR1-Related Myopathy
- PMID: 39409197
- PMCID: PMC11477233
- DOI: 10.3390/ijms251910867
Compound Heterozygous RYR1 Variants in a Patient with Severe Congenital Myopathy: Case Report and Comparison with Additional Cases of Recessive RYR1-Related Myopathy
Abstract
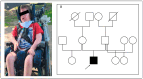
Pathogenic variants in the ryanodine receptor 1 (RYR1) gene are causative for a wide spectrum of muscular phenotypes, ranging from malignant hyperthermia over mild, non-progressive to severe congenital myopathy. Both autosomal dominant and recessive inheritance can occur, with the more severe forms usually showing recessive inheritance. However, genotype-phenotype correlations are complicated due to the large size of the gene and heterogeneous phenotypes. We present a 6-year-old patient with severe congenital myopathy, carrying a heterozygous pathogenic RYR1 variant inherited from the healthy mother. Through whole genome sequencing we identified a second, deep intronic RYR1 variant that has recently been described in another patient with severe congenital myopathy and shown to affect splicing. Segregation analyses confirmed the variants to be compound heterozygous. We compared our patient's phenotype to that of the patient from the literature as well as five additional patients with compound heterozygous RYR1 variants from our center. The main overlapping features comprised congenital onset, predominant muscular hypotonia, and normal creatine kinase (CK) levels, while overall clinical expression varied substantially. Interestingly, both patients carrying the new intronic splice variant showed a very severe disease course. More widespread use of genome sequencing will open the way for better genotype-phenotype correlations.
Keywords: RYR1-related myopathies; congenital myopathy; ryanodine receptor 1 (RYR1); splice variant; whole genome sequencing (WGS).
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


References
-
- Zecevic N., Arsenijevic V., Manolakos E., Papoulidis I., Theocharis G., Sartsidis A., Tsagas T., Tziotis I., Dagklis T., Kalogeros G., et al. New Compound Heterozygous Splice Site Mutations of the Skeletal Muscle Ryanodine Receptor (RYR1) Gene Manifest Fetal Akinesia: A Linkage with Congenital Myopathies. Mol. Syndromol. 2020;11:104–109. doi: 10.1159/000507034. - DOI - PMC - PubMed
-
- Lawal T.A., Todd J.J., Witherspoon J.W., Bönnemann C.G., Dowling J.J., Hamilton S.L., Meilleur K.G., Dirksen R.T. Ryanodine receptor 1-related disorders: An historical perspective and proposal for a unified nomenclature. Skelet. Muscle. 2020;10:32. doi: 10.1186/s13395-020-00243-4. - DOI - PMC - PubMed
-
- Klein A., Lillis S., Munteanu I., Scoto M., Zhou H., Quinlivan R., Straub V., Manzur A.Y., Roper H., Jeannet P.-Y., et al. Clinical and genetic findings in a large cohort of patients with ryanodine receptor 1 gene-associated myopathies. Hum. Mutat. 2012;33:981–988. doi: 10.1002/humu.22056. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

