Enhancing diffusion-weighted prostate MRI through self-supervised denoising and evaluation
- PMID: 39414914
- PMCID: PMC11484701
- DOI: 10.1038/s41598-024-75007-x
Enhancing diffusion-weighted prostate MRI through self-supervised denoising and evaluation
Abstract
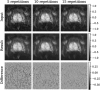
Diffusion-weighted imaging (DWI) is a magnetic resonance imaging (MRI) technique that provides information about the Brownian motion of water molecules within biological tissues. DWI plays a crucial role in stroke imaging and oncology, but its diagnostic value can be compromised by the inherently low signal-to-noise ratio (SNR). Conventional supervised deep learning-based denoising techniques encounter challenges in this domain as they necessitate noise-free target images for training. This work presents a novel approach for denoising and evaluating DWI scans in a self-supervised manner, eliminating the need for ground-truth data. By leveraging an adapted version of Stein's unbiased risk estimator (SURE) and exploiting a phase-corrected combination of repeated acquisitions, we outperform both state-of-the-art self-supervised denoising methods and conventional non-learning-based approaches. Additionally, we demonstrate the applicability of our proposed approach in accelerating DWI scans by acquiring fewer image repetitions. To evaluate denoising performance, we introduce a self-supervised methodology that relies on analyzing the characteristics of the residual signal removed by the denoising approaches.
© 2024. The Author(s).
Conflict of interest statement
L.P. and J.H. receive Ph.D. funding from Siemens Healthineers AG. O.D., F.W., E.W., T.B., C.E., D.N. and T.W. are employees of Siemens Healthineers AG. All other authors declare no competing interests.
Figures



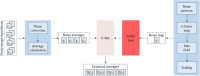


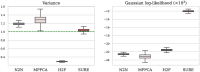
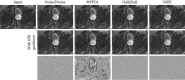


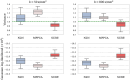

References
-
- Koh, D.-M. & Collins, D. J. Diffusion-weighted MRI in the body: applications and challenges in oncology. Am. J. Roentgenol. 188, 1622–1635 (2007). - PubMed
-
- Roberts, T. P. & Rowley, H. A. Diffusion weighted magnetic resonance imaging in stroke. Eur. J. Radiol. 45, 185–194 (2003). - PubMed
-
- Corcuera-Solano, I. et al. DWI of the prostate: Comparison of a faster diagonal acquisition to standard three-scan trace acquisition. J. Magn. Reson. Imaging 46, 1767–1775 (2017). - PubMed
-
- Ichikawa, T. et al. High-b-value diffusion-weighted MRI in colorectal cancer. Am. J. Roentgenol. 187, 181–184 (2006). - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

