This is a preprint.
A chronic Pseudomonas aeruginosa mouse lung infection modeling the pathophysiology and inflammation of human cystic fibrosis
- PMID: 39416002
- PMCID: PMC11482824
- DOI: 10.1101/2024.10.07.617039
A chronic Pseudomonas aeruginosa mouse lung infection modeling the pathophysiology and inflammation of human cystic fibrosis
Update in
-
A chronic Pseudomonas aeruginosa mouse lung infection modeling the mucus obstruction, lung function, and inflammation of human cystic fibrosis.Infect Immun. 2025 Jul 8;93(7):e0023025. doi: 10.1128/iai.00230-25. Epub 2025 Jun 13. Infect Immun. 2025. PMID: 40512037 Free PMC article.
Abstract
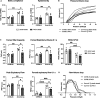
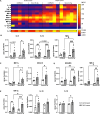
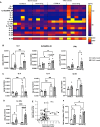
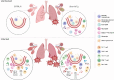
Investigation of chronic cystic fibrosis (CF) lung infections has been limited by a lack of murine models that reproduce obstructive lung pathology, chronicity of bacterial infections, and complex inflammation in human CF lung pathology. Three different approaches have been used separately to address these limitations, including using transgenic Scnn1b-Tg mice overexpressing a lung epithelial sodium channel to mimic the mucus-rich and hyperinflammatory CF lung environment, using synthetic CF sputum medium (SCFM) in an acute infection to induce bacterial phenotypes consistent with human CF, or using agar beads to promote chronic infections. Here, we combine these three models to establish a chronic Pseudomonas aeruginosa lung infection model using SCFM agar beads and Scnn1b-Tg mice (SCFM-Tg-mice) to recapitulate nutrients, mucus, and inflammation characteristic of the human CF lung environment. Like people with CF, SCFM-Tg-mice failed to clear bacterial infections. Lung function measurements showed that infected SCFM-Tg-mice had decreased inspiratory capacity and compliance, elevated airway resistance, and significantly reduced FVC and FEV0.1. Using spectral flow cytometry and multiplex cytokine arrays we show that, like people with CF, SCFM-Tg-mice developed inflammation characterized by eosinophil infiltration and Th2 lymphocytic cytokine responses. Chronically infected SCFM-Tg-mice developed an exacerbated mix of innate and Th1, Th2, and Th17-mediated inflammation, causing higher lung cellular damage, and elevated numbers of unusual Siglec F+ neutrophils. Thus, SCFM-Tg-mice represents a powerful tool to investigate bacterial pathogenesis and potential treatments for chronic CF lung infections and reveal a potential role for Siglec F+ neutrophils in CF inflammation.
Figures




Similar articles
-
A chronic Pseudomonas aeruginosa mouse lung infection modeling the mucus obstruction, lung function, and inflammation of human cystic fibrosis.Infect Immun. 2025 Jul 8;93(7):e0023025. doi: 10.1128/iai.00230-25. Epub 2025 Jun 13. Infect Immun. 2025. PMID: 40512037 Free PMC article.
-
Optimizing synthetic cystic fibrosis sputum media for growth of non-typeable Haemophilus influenzae.Access Microbiol. 2025 Jun 20;7(6):000979.v3. doi: 10.1099/acmi.0.000979.v3. eCollection 2025. Access Microbiol. 2025. PMID: 40548129 Free PMC article.
-
Antibiotic strategies for eradicating Pseudomonas aeruginosa in people with cystic fibrosis.Cochrane Database Syst Rev. 2017 Apr 25;4(4):CD004197. doi: 10.1002/14651858.CD004197.pub5. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2023 Jun 2;6:CD004197. doi: 10.1002/14651858.CD004197.pub6. PMID: 28440853 Free PMC article. Updated.
-
Non-invasive ventilation for cystic fibrosis.Cochrane Database Syst Rev. 2017 Feb 20;2(2):CD002769. doi: 10.1002/14651858.CD002769.pub5. Cochrane Database Syst Rev. 2017. PMID: 28218802 Free PMC article.
-
Exercise versus airway clearance techniques for people with cystic fibrosis.Cochrane Database Syst Rev. 2022 Jun 22;6(6):CD013285. doi: 10.1002/14651858.CD013285.pub2. Cochrane Database Syst Rev. 2022. PMID: 35731672 Free PMC article.
References
-
- Sanders D. B., Bittner R. C. L., Rosenfeld M., Redding G. J., Goss C. H., Pulmonary exacerbations are associated with subsequent FEV1 decline in both adults and children with cystic fibrosis. Pediatric Pulmonology 46, 393–400 (2011). - PubMed
-
- Vaillancourt M., Galdino A. C. M., Limsuwannarot S. P., Celedonio D., Dimitrova E., Broerman M., Bresee C., Doi Y., Lee J. S., Parks W. C., Jorth P., A compensatory RNase E variation increases Iron Piracy and Virulence in multidrug-resistant Pseudomonas aeruginosa during Macrophage infection. PLOS Pathogens 19, e1010942 (2023). - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
