Challenges and Opportunities for Consideration of Efavirenz Drug Repurposing for Alzheimer's Disease Therapeutics
- PMID: 39421657
- PMCID: PMC11480897
- DOI: 10.1021/acsptsci.4c00229
Challenges and Opportunities for Consideration of Efavirenz Drug Repurposing for Alzheimer's Disease Therapeutics
Abstract
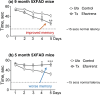
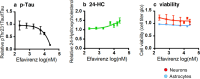
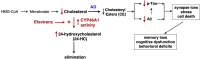
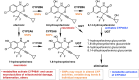
Therapeutic research and development for Alzheimer's disease (AD) has been an area of intense research to alleviate memory loss and neurodegeneration. There is growing interest in drug repositioning and repurposing strategies for FDA-approved medications as potential candidates that may further advance AD therapeutics. The FDA drug efavirenz has been investigated as a candidate drug for repurposing as an AD medication. The proposed mechanism of action of efavirenz (at low doses) is the activation of the neuron-specific enzyme CYP46A1 that converts excess brain cholesterol into 24-hydroxycholesterol (24-HC) that is exported to the periphery. Efavirenz at a low dose was found to improve memory deficit in the 5XFAD model of AD that was accompanied by elevated 24-HC and reduction in Aβ; furthermore, efavirenz reduced pTau and excess cholesterol levels in human iPSC-derived Alzheimer's neurons. The low dose of efavirenz used in the AD mouse model to increase 24-HC contrasts with the use of more than 100-fold higher doses of efavirenz for clinical treatment of human immunodeficiency virus (HIV) through inhibition of reverse transcriptase. Low doses of efavirenz may avoid neurotoxic adverse effects that occur at high efavirenz doses used for HIV treatment. This review evaluates the drug properties of efavirenz with respect to its preclinical data on regulating memory deficit, pharmacokinetics, pharmacodynamics, metabolites, and genetic variabilities in drug metabolism as well as its potential adverse effects. These analyses discuss the challenges and questions that should be addressed in future studies to consider the opportunity for low dose efavirenz as a candidate for AD drug development.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
