Disrupting stroke-induced GAT-1-syntaxin1A interaction promotes functional recovery after stroke
- PMID: 39423810
- PMCID: PMC11604526
- DOI: 10.1016/j.xcrm.2024.101789
Disrupting stroke-induced GAT-1-syntaxin1A interaction promotes functional recovery after stroke
Abstract
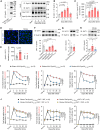
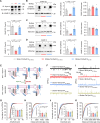
Although stroke is a frequent cause of permanent disability, our ability to promote stroke recovery is limited. Here, we design a small-molecule stroke recovery promoting agent that works by dissociating γ-aminobutyric acid (GABA) transporter 1 (GAT-1) from syntaxin1A (Synt1A), a soluble N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE) protein. Stroke induces an increase in GAT-1-Synt1A interaction in the subacute phase, a critical period for functional recovery. Uncoupling GAT-1-Synt1A reverses stroke-induced GAT-1 dysfunction and cortical excitability decline and enhances synaptic GABAergic inhibition and consequently cortical oscillations and network plasticity by facilitating the assembly of the SNARE complex at the synapse. Based on the molecular mechanism of GAT-1 binding to Synt1A, we design GAT-1-Synt1A blockers. Among them, ZLQ-3 exhibits the greatest potency. Intranasal use of ZLQ-3-1, a glycosylation product of ZLQ-3, substantially lessens impairments of sensorimotor and cognitive functions in rodent models. This compound, or its analogs, may serve as a promoting agent for stroke recovery.
Keywords: GAT-1; functional recovery; network plasticity; stroke; syntaxin1A.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests D.-Y.Z., Y.-H.L., Y.-J.Q., and T.-Y.L. are inventors on the following PCT on ZLQ series compounds application: filed on 15/08/2024, application no. PCT/CN2024/112442. Y.-H.L., Y.-J.Q., and T.-Y.L. are inventors on the following Chinese patent on ZLQ series compounds application: publication no. CN117384253A.
Figures





References
-
- Hobeanu C., Lavallée P.C., Charles H., Labreuche J., Albers G.W., Caplan L.R., Donnan G.A., Ferro J.M., Hennerici M.G., Molina C.A., et al. Risk of subsequent disabling or fatal stroke in patients with transient ischaemic attack or minor ischaemic stroke: an international, prospective cohort study. Lancet Neurol. 2022;21:889–898. doi: 10.1016/S1474-4422(22)00302-7. - DOI - PubMed
-
- Suzuki K., Matsumaru Y., Takeuchi M., Morimoto M., Kanazawa R., Takayama Y., Kamiya Y., Shigeta K., Okubo S., Hayakawa M., et al. Effect of mechanical thrombectomy without vs with intravenous thrombolysis on functional outcome among patients with acute ischemic stroke: the SKIP randomized clinical trial. JAMA. 2021;325:244–253. doi: 10.1001/jama.2020.23522. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

