Efficacy and safety of topical glycopyrronium bromide in treating axillary hyperhidrosis: systematic review and meta-analysis
- PMID: 39424822
- PMCID: PMC11489785
- DOI: 10.1038/s41598-024-74430-4
Efficacy and safety of topical glycopyrronium bromide in treating axillary hyperhidrosis: systematic review and meta-analysis
Abstract
Background: Hyperhidrosis (HH), characterized by excessive sweating, poses a significant challenge to patients' quality of life. This meta-analysis evaluates the safety and efficacy of topical glycopyrronium bromide (GBP) in treating primary hyperhidrosis, a chronic condition affecting various body regions. Despite its prevalence, primary axillary hyperhidrosis is often undertreated due to a lack of awareness and social stigma.
Methods: Following PRISMA guidelines, we conducted a systematic review and meta-analysis of randomized controlled trials comparing GBP to a placebo in primary hyperhidrosis patients. Eligibility criteria included outcomes related to perspiration suppression and symptom improvement.
Results: Four RCTs involving 1401 patients were included. GBP significantly increased Hyperhidrosis Disease Severity Scale (HDSS) responders (RR = 2.33, 95% CI [1.99 to 2.74], p < 0.00001) and Axillary Sweating Daily Diary (ASDD/ASDD-C) responders (MD = 3.07, 95% CI [2.32 to 4.06], p < 0.002) without significantly causing adverse events. Dermatology life quality index was also significantly improved in the GBP group (MD = -2.32, 95% CI [-3.09, -1.55], P < 0.00001).
Conclusion: GBP demonstrated effectiveness in reducing sweat production while improving HDSS and DLQI scores. Adverse events included dry mouth and anticholinergic effects. Dry eye and local skin reactions were not significant, which makes GBP promising in managing primary hyperhidrosis, offering improvements in symptoms and quality of life. While adverse events should be considered, further research with larger sample sizes and long-term follow-up is warranted for comprehensive clinical integration.
Keywords: Adverse events; Glycopyrronium Bromide; Hyperhidrosis; Meta-analysis; Quality of life.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures









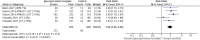
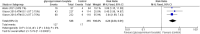


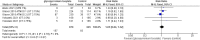
References
-
- Hyun, M. Y. et al. Efficacy and safety of topical glycopyrrolate in patients with facial hyperhidrosis: a randomized, multicentre, double-blinded, placebo‐controlled, split‐face study. J. Eur. Acad. Dermatol. Venereol.29, 278–282 (2015). - PubMed
-
- Glaser, D. A. et al. Topical glycopyrronium tosylate for the treatment of primary axillary hyperhidrosis: results from the ATMOS-1 and ATMOS-2 phase 3 randomized controlled trials. J. Am. Acad. Dermatol.80, 128–138e2 (2019). - PubMed
-
- Szeimies, R. et al. Long‐term efficacy and safety of 1% glycopyrronium bromide cream in patients with severe primary axillary hyperhidrosis: results from a phase 3b trial. J. Eur. Acad. Dermatol. Venereol.37, 823–830 (2023). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

