Alpha-1-B glycoprotein (A1BG) inhibits sterol-binding and export by CRISP2
- PMID: 39433128
- PMCID: PMC11599453
- DOI: 10.1016/j.jbc.2024.107910
Alpha-1-B glycoprotein (A1BG) inhibits sterol-binding and export by CRISP2
Abstract
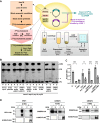
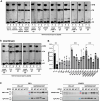
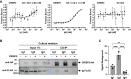
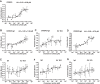
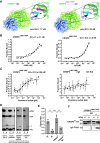
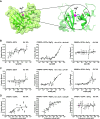
Proteins belonging to the CAP superfamily are present in all kingdoms of life and have been implicated in various processes, including sperm maturation and cancer progression. They are mostly secreted glycoproteins and share a unique conserved CAP domain. The precise mode of action of these proteins, however, has remained elusive. Saccharomyces cerevisiae expresses three members of this protein family, which bind sterols in vitro and promote sterol secretion from cells. This sterol-binding and export function of yeast Pry proteins is conserved in the mammalian cysteine-rich secretory protein (CRISP) proteins and other CAP superfamily members. CRISP3 is an abundant protein of the human seminal plasma and interacts with alpha-1-B glycoprotein (A1BG), a human plasma glycoprotein that is upregulated in different types of cancers. Here, we examined whether the interaction between CRISP proteins and A1BG affects the sterol-binding function of CAP family members. Coexpression of A1BG with CAP proteins abolished their sterol export function in yeast and their interaction inhibits sterol-binding in vitro. We map the interaction between A1BG and CRISP2 to the third of five repeated immunoglobulin-like domains within A1BG. Interestingly, the interaction between A1BG and CRISP2 requires magnesium, suggesting that coordination of Mg2+ by the highly conserved tetrad residues within the CAP domain is essential for a stable interaction between the two proteins. The observation that A1BG modulates the sterol-binding function of CRISP2 has potential implications for the role of A1BG and related immunoglobulin-like domain containing proteins in cancer progression and the toxicity of reptile venoms containing CRISP proteins.
Keywords: CAP protein superfamily; Saccharomyces cerevisiae, snake venom toxins; cysteine-rich secretory protein (CRISP); immunoglobulin domain (Ig); oprin; protein-protein interactions; sterol export.
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflict of interest with the content of this article.
Figures


References
-
- Gibbs G.M., Roelants K., O’Bryan M.K. The CAP superfamily: cysteine-rich secretory proteins, antigen 5, and pathogenesis-related 1 proteins--roles in reproduction, cancer, and immune defense. Endocr. Rev. 2008;29:865–897. - PubMed
-
- Schneiter R., Di Pietro A. The CAP protein superfamily: function in sterol export and fungal virulence. Biomol. Concepts. 2013;4:519–525. - PubMed
-
- Szyperski T., Fernandez C., Mumenthaler C., Wuthrich K. Structure comparison of human glioma pathogenesis-related protein GliPR and the plant pathogenesis-related protein P14a indicates a functional link between the human immune system and a plant defense system. Proc. Natl. Acad. Sci. U. S. A. 1998;95:2262–2266. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

