Eye on the horizon: The metabolic landscape of the RPE in aging and disease
- PMID: 39433211
- PMCID: PMC11833275
- DOI: 10.1016/j.preteyeres.2024.101306
Eye on the horizon: The metabolic landscape of the RPE in aging and disease
Abstract
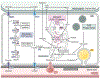
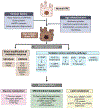
To meet the prodigious bioenergetic demands of the photoreceptors, glucose and other nutrients must traverse the retinal pigment epithelium (RPE), a polarised monolayer of cells that lie at the interface between the outer retina and the choroid, the principal vascular layer of the eye. Recent investigations have revealed a metabolic ecosystem in the outer retina where the photoreceptors and RPE engage in a complex exchange of sugars, amino acids, and other metabolites. Perturbation of this delicate metabolic balance has been identified in the aging retina, as well as in age-related macular degeneration (AMD), the leading cause of blindness in the Western world. Also common in the aging and diseased retina are elevated levels of cytokines, oxidative stress, advanced glycation end-products, increased growth factor signalling, and biomechanical stress - all of which have been associated with metabolic dysregulation in non-retinal cell types and tissues. Herein, we outline the role of these factors in retinal homeostasis, aging, and disease. We discuss their effects on glucose, mitochondrial, lipid, and amino acid metabolism in tissues and cell types outside the retina, highlighting the signalling pathways through which they induce these changes. Lastly, we discuss promising avenues for future research investigating the roles of these pathological conditions on retinal metabolism, potentially offering novel therapeutic approaches to combat age-related retinal disease.
Keywords: Age-related macular degeneration; Metabolic ecosystem; RPE aging; RPE metabolism; Retina inflammation; Retina metabolism; Retinal pigment epithelium.
Copyright © 2024 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures









References
-
- Abe Y, Sakairi T, Beeson C, Kopp JB, 2013. TGF-β1 stimulates mitochondrial oxidative phosphorylation and generation of reactive oxygen species in cultured mouse podocytes, mediated in part by the mTOR pathway. Am. J. Physiol. Ren. Physiol 305, 1477–1490. 10.1152/ajprenal.00182.2013. - DOI - PMC - PubMed
-
- Ach T, Tolstik E, Messinger JD, Zarubina AV, Heintzmann R, Curcio CA, 2015. Lipofuscin redistribution and loss accompanied by cytoskeletal stress in retinal pigment epithelium of eyes with age-related macular degeneration. Investig. Ophthalmol. Vis. Sci 56, 3242–3252. 10.1167/iovs.14-16274. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

