Distinct immunomodulation elicited by young versus aged extracellular vesicles in bone marrow-derived macrophages
- PMID: 39434100
- PMCID: PMC11492788
- DOI: 10.1186/s12979-024-00472-x
Distinct immunomodulation elicited by young versus aged extracellular vesicles in bone marrow-derived macrophages
Abstract
Background: Previous research has indicated that extracellular vesicles (EVs) potentially play significant roles in multiple ageing phenotypes. This study uses a factorial experimental design to explore the interactions between circulating EVs and bone marrow-derived macrophages (BMDMs) isolated from young (7-12 weeks) and aged (70-90 weeks) mice.
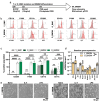
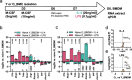
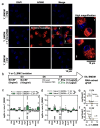
Results: In this study, plasma EVs from young (Y_EV) and aged (O_EV) mice were isolated and compared based on abundance, size, and miRNA cargo. Compared to some previous studies, we found relatively few differences in EV miRNA cargo between Y_EVs and O_EVs. Young and old EVs were then used to stimulate naïve BMDMs isolated from young (Y_BMDM) and aged (O_BMDM) mice. A panel of five "M1" and six "M2" macrophage markers were used to assess the degree of polarisation. Our results revealed differences in the immunomodulatory effects of Y_EVs and O_EVs in Y_BMDMs and O_BMDMs. Y_EVs induced less pro-inflammatory gene expression, while O_EVs exhibited a more varied impact, promoting both pro- and anti-inflammatory markers. However, neither EV population induced a clearly defined 'M1' or 'M2' macrophage phenotype. We also report that EVs elicited responses that differed markedly from those induced by whole plasma. Plasma from old mice had strong pro-inflammatory effects on Y_BMDMs, increasing Il1b, Nlrp3 and Tnfa. However, O_EVs did not have these effects, supporting current evidence that EVs are a separate component of circulating factors during ageing. More research is needed to elucidate specific factors involved in inflammageing processes.
Conclusions: Our findings reveal age-related differences in EV cargo and function, with young EVs tending to suppress inflammatory markers more effectively than aged EVs. However, this is not straightforward, and EVs often promoted both M1 and M2 markers. These results suggest that EVs are a distinct component of circulating factors and hold potential for therapeutic strategies aimed at mitigating age-related inflammation and immune dysregulation.
Keywords: Ageing; Aging; Blood plasma; Cytokine; Exosome; Inflammation; Macrophage; miRNA.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Grants and funding
- NSTC 113-2314-B-038-035-MY3/National Science and Technology Council
- 111-TMU023, 112-TMU082/Taipei Medical University
- 111-TMU023, 112-TMU082/Taipei Medical University
- 111TMU-SHH-19, 113TMU-SHH-26/Taipei Medical University-Shuang Ho Hospital, Ministry of Health and Welfare
- 111TMU-SHH-19, 113TMU-SHH-26/Taipei Medical University-Shuang Ho Hospital, Ministry of Health and Welfare
LinkOut - more resources
Full Text Sources

