Preferences for oral and injectable PrEP among qualitative sub-study participants in HPTN 084
- PMID: 39441777
- PMCID: PMC11498703
- DOI: 10.1371/journal.pone.0309811
Preferences for oral and injectable PrEP among qualitative sub-study participants in HPTN 084
Abstract
Background: HPTN 084 compared the safety and efficacy of long-acting injectable cabotegravir (CAB) to daily oral TDF/FTC for prevention of HIV-1 in uninfected African women. Like a similar trial in MSM/TGW (HPTN 083), the trial was stopped early for efficacy, expediting the need to consider introduction strategies for different populations. We examine survey and qualitative data from a four-country sub-study to examine oral and injectable PrEP acceptability and considerations for CAB access among African women.
Methods: Participants completed baseline and follow-up surveys on HIV risk perception, sexual behavior. product acceptability and adherence during the blinded trial. Additionally, up to two in-depth interviews each with 73 sub-study participants explored product use and trial-related experiences, during the blinded and unblinded study periods. Using survey data, we classified participants as: engaged in female sex work (FSW), having multiple non-transactional partners, or monogamous. A study statistician identified participants' assigned study arm. We followed a thematic analysis process to read transcripts, develop a codebook and apply codes in NVivo to transcripts with intermittent intercoder reliability checks; using Excel matrices to explore differences across risk categories and study arms.
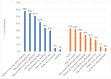
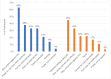
Findings: Participants overwhelmingly preferred injections to pills, appreciating the ease, convenience, and privacy of a long-acting formulation. Many participants described challenges with contraceptive and/or study pill adherence, impeded by late night work, unexpected travel, or heavy drinking. Women in the TDF/FTC arm were more likely to describe side effects, compared to those in the CAB arm. Pain also varied widely by study arm. When considering post-trial access to CAB, limited PrEP knowledge, cost and concerns around stigma and poor service quality were potential access barriers.
Conclusion: Women's desire for privacy and ease of use outweighed injectable concerns, resulting in a strong preference for CAB. Cost and accessibility will need to be addressed by implementation programs.
Copyright: © 2024 Tolley et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
References
-
- UNAIDS. Confronting inequalities: lessons for pandemic responses from 40 years of AIDS. 2021.
-
- CDC Statement on FDA Approval of Drug for HIV Prevention [Internet]. Atlanta, GA; 2012 [cited April 13, 2022]. https://www.cdc.gov/nchhstp/newsroom/2012/FDA-ApprovesDrugStatement.html
-
- World Health Organization. Guildeline on When to Start Antiretroviral Therapy and on Pre-Exposure Prophylaxis for HIV. Geneva, Switzerland: 2015. - PubMed
-
- Organization WH. Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection: recommendations for a public health approach. Switzerland: WHO, 2016. - PubMed
-
- FDA approves second drug to prevent HIV infection as part of ongoing efforts to end the HIV epiidemic [Internet]. 2019 [cited April 13, 2022]. https://www.fda.gov/news-events/press-announcements/fda-approves-second-...
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

