The Influence of the Estrous Cycle on Neuropeptide S Receptor-Mediated Behaviors
- PMID: 39443144
- PMCID: PMC11585311
- DOI: 10.1124/jpet.124.002415
The Influence of the Estrous Cycle on Neuropeptide S Receptor-Mediated Behaviors
Abstract
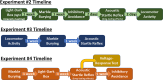
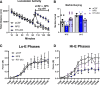
The neuropeptide S receptor (NPSR) has been identified as a potential therapeutic target for anxiety and post-traumatic stress disorder. Central administration of neuropeptide S (NPS) in male mice produces anxiolytic-like effects, hyperlocomotion, and memory enhancement. Currently, the literature is limited in the number of studies investigating the effects of NPS in female test subjects despite females facing a higher prevalence of anxiety-related pathology, as well as greater risk for adverse effects while taking psychoactive drugs. Moreover, no previous studies have considered the influence of estrous cycle on the effects of NPS. The present study investigates whether NPS-mediated behavioral phenotypes seen in males translate to females, and whether they are affected by estrous cycle stage. Female C57BL/6NCr mice were intracerebroventricularly cannulated and underwent behavioral paradigms to test locomotion, anxiety, and memory. Estrous cycle stage was determined through examination of vaginal cytology. Our results provide evidence that NPS-mediated behaviors are influenced by the estrous cycle. Administration of NPS decreased anxiety-like behaviors more robustly when the female mice were in high estrogen stages of the estrous cycle. Therefore, the desired anxiolytic-like effects of targeting the NPSR are intact in female mice. However, these effects may to be influenced by the stage of the estrous cycle. The NPSR remains a strong potential drug target for new anxiolytic compounds and based on our initial observations further studies exploring the interaction of estrous cycle and the NPS system are warranted. SIGNIFICANCE STATEMENT: The neuropeptide S (NPS) receptor has been identified as a potential target for treating anxiety, a condition that is most prevalent in females. Therefore, the potential interaction of estrous cycle with the NPS system described in the present study is an important first step in understanding the function of the NPS system in females.
U.S. Government work not protected by U.S. copyright.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures



References
-
- Badia-Elder NE, Henderson AN, Bertholomey ML, Dodge NC, Stewart RB (2008) The effects of neuropeptide S on ethanol drinking and other related behaviors in alcohol-preferring and -nonpreferring rats. Alcohol Clin Exp Res 32:1380–1387. - PubMed
-
- Clark SD, Duangdao DM, Schulz S, Zhang L, Liu X, Xu Y-L, Reinscheid RK (2011) Anatomical characterization of the neuropeptide S system in the mouse brain by in situ hybridization and immunohistochemistry. J Comp Neurol 519:1867–1893. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
