A unique and biocompatible corneal collagen crosslinking in vivo
- PMID: 39443505
- PMCID: PMC11500088
- DOI: 10.1038/s41598-024-71871-9
A unique and biocompatible corneal collagen crosslinking in vivo
Erratum in
-
Author Correction: A unique and biocompatible corneal collagen crosslinking in vivo.Sci Rep. 2025 May 30;15(1):19046. doi: 10.1038/s41598-025-01649-0. Sci Rep. 2025. PMID: 40447612 Free PMC article. No abstract available.
Abstract
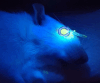
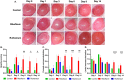
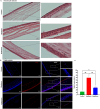
Corneal crosslinking (CXL) is a widely applied technique to halt the progression of ectatic diseases through increasing the thickness and mechanical stiffness of the cornea. This study investigated the biocompatibility and efficiency of a novel CXL procedure using ruthenium and blue light in rat corneas and evaluated parameters important for clinical application. To perform the CXL procedure, the corneal epithelium of rats was removed under anaesthesia, followed by the application of a solution containing ruthenium and sodium persulfate (SPS). The corneas were then exposed to blue light at 430 nm at 3 mW/cm2 for 5 min. Rat corneas were examined and evaluated for corneal opacity, corneal and limbal neovascularization, and corneal epithelial regeneration on days 0, 1, 3, 6, 8, and 14. On day 28, the corneas were isolated for subsequent tissue follow-up and analysis. CXL with ruthenium and blue light showed rapid epithelial healing, with 100% regeneration of the corneal epithelium and no corneal opacity on day 6. The ruthenium group also exhibited significantly reduced corneal (p < 0.01) and limbal neovascularization (p < 0.001). Histological analysis revealed no signs of cellular damage or apoptosis, which further confirms the biocompatibility and nontoxicity of our method. Confocal and scanning electron microscopy (SEM) images confirmed high density of collagen fibrils, indicating efficient crosslinking and enhanced structural integrity. This study is unique that demonstrates in vivo safety, biocompatibility, and functionality of ruthenium and blue light CXL. This approach can prevent toxicity caused by UV-A light and can be an immediate alternative compared to the existing crosslinking procedures that have side effects and clinical risks for the patients.
Keywords: Blue light; Collagen; Cornea; Crosslinking; In vivo; Ruthenium.
© 2024. The Author(s).
Conflict of interest statement
The authors S.K., A.Ş., A.Y., and A.G. are inventors and have filled a patent through the Koc University (A METHOD AND A PHARMACEUTICAL COMPOSITION FOR VISIBLE LIGHT-INDUCED CORNEAL CROSSLINKING WITH RUTHENIUM COMPOUNDS FOR THE TREATMENT OF EYE DISORDERS, application number PCT/TR2022/050954, in process) for the use of ruthenium for corneal collagen crosslinking. All the remaining authors declare no conflict of interest.
Figures



References
-
- MacAdam, A. J. etal. Low energy blue pulsed light-activated injectable materials for restoring thinning corneas, advanced functional materials (2023).
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

