Early adoption of triamcinolone acetonide suprachoroidal injection for uveitic macular edema: a physician survey
- PMID: 39444009
- PMCID: PMC11515593
- DOI: 10.1186/s13104-024-06969-4
Early adoption of triamcinolone acetonide suprachoroidal injection for uveitic macular edema: a physician survey
Abstract
Objective: To obtain physicians' "real-world" perspectives on early experiences with triamcinolone acetonide suprachoroidal injection (SCS-TA) for treatment of patients with uveitic macular edema (UME).
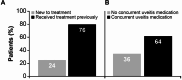
Results: Twelve retina/uveitis specialists in the United States were surveyed about SCS-TA injection procedure and patient outcomes. Survey participants administered ≥ 291 SCS-TA injections to 243 patients with UME with various disease characteristics (etiologies, chronicity, and anatomical subtypes). Commonly reported reasons for SCS-TA adoption included potential for lowering the risk of steroid-associated intraocular pressure elevations versus intravitreal injections or implants (100%), potential for longer duration of action versus intravitreal steroid injections or implants (92%), and desire to use a new delivery modality (83%). Nearly all participants (92%) found injection procedure relatively easy post-training, with most (75%) procedurally comfortable after completing 2-5 injections. 58% of participants indicated that their patients gained 2-3 lines of vision by first follow-up visit, and 92% reported having patients who experienced 100-150 μm or greater reduction in central subfield thickness. Overall, 92% of participants were satisfied with SCS-TA treatment outcomes. Findings from this survey of early adopters of SCS-TA indicate that the suprachoroidal injection technique was easy to learn and resulted in favorable patient outcomes consistent with clinical trial data.
Keywords: Macular edema; Retina; Suprachoroidal injection; Survey; Treatment; Uveitis.
© 2024. The Author(s).
Conflict of interest statement
Competing interests: CRH reports being a consultant for Allergan, Alimera, Bausch + Lomb, Clearside Biomedical, Inc., and EyePoint Pharmaceuticals. SDW reports being a speaker for Apellis, Bausch + Lomb, Genentech/Roche, Regeneron, and Spark Therapeutics. SDW reports being a consultant for Allergan, Astellis/Iveric Bio, Bausch + Lomb, Castle Biosciences, EyePoint Pharmaceuticals/Alimera, Genentech/Roche, Ideaya Biosciences, Lupin, Novartis, and Regeneron. PEN reports receiving research and consulting fees from EyePoint Pharmaceuticals and Genentech. PEN is supported by the Knights Templar Eye Foundation, Inc. Career Starter grant. MAS reports receiving consulting fees from Aerie, Alimera, Allegro, Allergan, Eyepoint, Genentech, Kodiak, Novartis, Regeneron, and Santen; serving on the speakers’ bureau for Allergan, Apellis, Genentech, Mallinckrodt, Novartis, Regeneron, and Spark; and performing contracted research for Aerie, Allegro, Allergan, DRCR, Genentech, Icon, Ionis, KalVista, Kodiak, Novartis, Opthea, Optos, Regeneron, Santen, Senju, and Sydnexis. Survey participants (CRH, SDW, PYC, DJW, PEN, KJB, MSD, DSC, VRR, LMR, MS, and MAS) received an honorarium from Bausch & Lomb Americas, Inc. TB and MY are employees of Bausch + Lomb, Bridgewater, NJ.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

