Fentanyl enhances immune cell response through TLR4/MD-2 complex
- PMID: 39444612
- PMCID: PMC11496304
- DOI: 10.3389/fphar.2024.1468644
Fentanyl enhances immune cell response through TLR4/MD-2 complex
Abstract
Introduction: Opioids have been shown to induce neuroinflammation and immune cell activation, that might contribute to some of the opioid side effects, such as opioid-induced tolerance and paradoxical hyperalgesia. In this context, TLR4/MD-2 complex has been proposed as an off-target site for opioid action. This study was aimed at investigating the effect of fentanyl on lipopolysaccharide (LPS)-induced TLR4/MD-2 activation in rat primary microglia and human monocyte-derived macrophages (MDM).
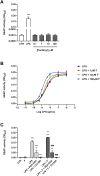
Materials and methods: The effect of fentanyl was first explored by measuring the expression and release of different proinflammatory mediators in primary rat microglia and human MDM by real-time PCR and ELISA. Then, the involvement of TLR4/MD-2 signaling was investigated studying NF-κB activation in HEK293 cells stably transfected with human TLR4, MD-2, and CD14 genes (HEK-Blue hTLR4 cells) and in human MDM.
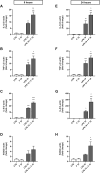
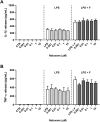
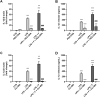
Results: Fentanyl increased mRNA levels, as well as the LPS-induced secretion of proinflammatory mediators in primary microglia and MDM. Two inhibitors of TLR4/MD-2 signaling, namely the oxazoline derivative of N-palmitoylethanolamine (PEA-OXA) and CLI-095, blocked the production and release of proinflammatory cytokines by microglia stimulated with LPS and fentanyl, suggesting that TLR4/MD-2 could be the target of the proinflammatory activity of fentanyl. Finally, we showed that fentanyl in combination with LPS activated NF-κB signaling in human MDM and in HEK-Blue hTLR4 cells and this effect was blocked by inhibitors of TLR4/MD-2 complex.
Discussion: These results provide new insight into the mechanism of the proinflammatory activity of fentanyl, which involves the activation of TLR4/MD-2 signaling. Our findings might facilitate the development of novel inhibitors of TLR4/MD-2 signaling to combine with opioid-based analgesics for effective and safe pain management.
Keywords: TLR4/MD-2 complex; fentanyl; inflammatory cytokines; macrophages; microglia.
Copyright © 2024 Chemello, Facci, Marcolin, Ramaschi, Barbierato, Giusti, Bolego and Zusso.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures


References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

