Fluid biomarkers in multiple sclerosis: from current to future applications
- PMID: 39444698
- PMCID: PMC11496979
- DOI: 10.1016/j.lanepe.2024.101009
Fluid biomarkers in multiple sclerosis: from current to future applications
Abstract
Multiple sclerosis (MS) is an immune-mediated inflammatory and degenerative disorder of the central nervous system (CNS) with heterogeneous clinical manifestations. In the last decade, the landscape of cerebrospinal fluid (CSF) and blood biomarkers as potential key tools for MS diagnosis, prognosis and treatment monitoring has evolved considerably, alongside magnetic resonance imaging (MRI). CSF analysis has the potential not only to provide information on the underlying immunopathology of the disease and exclude differential diagnoses, but also to predict the risk of future relapses and disability accrual, guide therapeutic decisions and thus improve patient outcomes. This Series article overviews the biological framework and current applicability of fluid biomarkers for MS, exploring their potential role in the molecular characterisation of the disease. We discuss recent advances in the field of neurochemistry that enabled the detection of brain-derived proteins in blood, opening the door to much more efficient longitudinal disease monitoring. Furthermore, we identify the current challenges in the application of fluid biomarkers for MS in a real-world setting, while offering recommendations for harnessing their full potential as key paraclinical tools to improve patient management and personalise treatment.
Keywords: Biomarkers; Blood; Cerebrospinal fluid; Multiple sclerosis.
© 2024 The Authors.
Conflict of interest statement
Massimiliano Di Filippo participated on advisory boards and steering committees for and received speaker or writing honoraria, research support and funding for travelling from Alexion, BMS, Bayer, Biogen Idec, Genzyme, Horizon, Janssen, Merck, Mylan, Novartis, Roche, Siemens Healthineers, Teva and Viatris. Lorenzo Gaetani participated on advisory boards for, and received writing honoraria and travel grants from Almirall, Biogen, Euroimmun, Fujirebio, Lilly, Merck, Mylan, Novartis, Roche, Sanofi, Siemens Healthineers and Teva. Diego Centonze is an advisory board member of Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck Serono, Novartis, Roche, Sanofi-Genzyme, and Teva, and received honoraria for speaking or consultation fees from Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck Serono, Novartis, Roche, Sanofi-Genzyme, and Teva. He is also the principal investigator in clinical trials for Bayer Schering, Biogen, Merck Serono, Mitsubishi, Novartis, Roche, Sanofi-Genzyme, and Teva. His preclinical and clinical research was supported by grants from Bayer Schering, Biogen Idec, Celgene, Merck Serono, Novartis, Roche, Sanofi-Genzyme and Teva. Harald Hegen has participated in meetings sponsored by, received speaker honoraria or travel funding from Bayer, Biogen, Bristol Myers Squibb, Horizon, Janssen, Merck, Novartis, Sanofi-Genzyme, Siemens, Teva, and received honoraria for acting as consultant for Biogen, Bristol Myers Squibb, Novartis, Roche, Sanofi-Genzyme and Teva. He is associate editor of Frontiers in Neurology. Jens Kuhle received speaker fees, research support, travel support, and/or served on advisory boards by Swiss MS Society, Swiss National Research Foundation (320030_189140/1), University of Basel, Progressive MS Alliance, Alnylam, Bayer, Biogen, Bristol Myers Squibb, Celgene, Immunic, Merck, Neurogenesis, Novartis, Octave Bioscience, Quanterix, Roche, Sanofi, Stata DX. Charlotte E. Teunissen performed contract research for Acumen, ADx Neurosciences, AC-Immune, Alamar, Aribio, Axon Neurosciences, Beckman–Coulter, BioConnect, Bioorchestra, Brainstorm Therapeutics, Celgene, Cognition Therapeutics, EIP Pharma, Eisai, Eli Lilly, Fujirebio, Grifols, Instant Nano Biosensors, Merck, Novo Nordisk, Olink, PeopleBio, Quanterix, Roche, Toyama, Vivoryon. She is editor in chief of Alzheimer Research and Therapy, and serves on editorial boards of Medidact Neurologie/Springer, and Neurology: Neuroimmunology & Neuroinflammation. She had speaker contracts for Eli Lilly, Grifols, Novo Nordisk, Olink and Roche. Mar Tintoré has received compensation for consulting services, speaking honoraria and research support from Almirall, Bayer Schering Pharma, Biogen-Idec, Genzyme, Janssen, Merck-Serono, Novartis, Roche, Sanofi-Aventis, Viela Bio and Teva Pharmaceuticals. Data Safety Monitoring Board for Parexel and UCB Biopharma, Relapse Adjudication Committee for IMCYSE SA. Luisa Maria Villar participated on advisory boards and for and received speaker or writing honoraria, research support and funding for travelling from BMS, Bayer, Binding Site, Biogen, Horizon, Janssen, Merck, Novartis, Roche, and Sanofi. Eline Willemse declared no competing interests. Henrik Zetterberg has served at scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZPath, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Merry Life, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave, has given lectures in symposia sponsored by Alzecure, Biogen, Cellectricon, Fujirebio, Lilly, and Roche, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work). Lucilla Parnetti declared no competing interests.
Figures
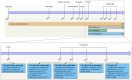

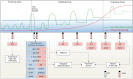
References
-
- Portaccio E., Magyari M., Havrdova E.K., et al. Multiple sclerosis: emerging epidemiological trends and redefining the clinical course. Lancet Reg Health Eur. 2024;44:100977. doi: 10.1016/j.lanepe.2024.100977. - DOI
-
- Thompson A.J., Banwell B.L., Barkhof F., et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17:162–173. - PubMed
-
- Rocca M.A., Preziosa P., Barkhof F., et al. MRI in the diagnosis and prognosis of multiple sclerosis. Lancet Reg Heal Eur. 2024;44:100978. doi: 10.1016/j.lanepe.2024.100978. - DOI
Publication types
LinkOut - more resources
Full Text Sources

