Pro re nata Treatment of Diabetic Macular Edema with Cycles of Three Injections of Anti-vascular Endothelial Growth Factor Injections
- PMID: 39444996
- PMCID: PMC11495293
- DOI: 10.4103/meajo.meajo_17_22
Pro re nata Treatment of Diabetic Macular Edema with Cycles of Three Injections of Anti-vascular Endothelial Growth Factor Injections
Abstract
Purpose: The purpose of this study was to report the visual and anatomic results at 12 and 24 months using the protocol of 3 monthly pro re nata (PRN) injections for diabetic macular edema (DME).
Methods: This was a retrospective chart review of 97 eyes with DME treated with a protocol of cycles consisting of 3 monthly injections of anti-vascular endothelial growth factor drugs on a PRN basis. Change in visual acuity was the main outcome measure. Macular thickness, number of injections, cycles, and visits in years 1 and 2 of follow-up were secondary outcomes.
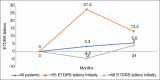
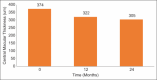
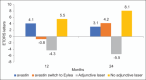
Results: Ninety-six patients with a mean age of 60.9 ± 9.96 years were followed for a mean of 22.17 ± 12.30 months. Ninety-two (95.9%), 3 (3%), and 1 (1%) patients were started on bevacizumab, ranibizumab, and aflibercept, respectively. Of bevacizumab patients, 17 (18.2%) were eventually switched to aflibercept. The mean 12-month improvement (standard deviation [SD]) was + 3.3 (17.4) letters (95% confidence interval [CI] = +0.36-+7.05, P < 0.001) after an average (SD) of 5.97 (2.98) injections over a mean (SD) of 1.7 (1.0) three-injection cycles. The mean 24-month improvement (SD) was + 5.6 (13.0) letters (95% CI: -0.28-11.05, P = 0.0186) after an average (SD) of 8.72 (6.31) injections over a mean (SD) of 2.9 (2.1) three-injection cycles. The mean central macular thickness (SD) at baseline, 12 months, and 24 months was 374 ± 120, 322 ± 88, and 305 ± 70 µm. Optical coherence tomography was fluid free at 12 and 24 months in 27.6% and 46% of eyes, respectively.
Conclusion: Comparable to real-world studies, this protocol can stabilize or improve vision in more than 85% of DME patients over 24 months. The most important factor in improvement of vision is increasing number of injections and visits.
Keywords: Anti-vascular endothelial growth factor; diabetic macular edema; intravitreal injections.
Copyright: © 2024 Middle East African Journal of Ophthalmology.
Conflict of interest statement
There are no conflicts of interest.
Figures
References
-
- Centers for Disease Control and Prevention . Atlanta, GA: Centers for Disease Control and Prevention, U.S. Dept of Health and Human Services; 2020. National Diabetes Statistics Report, 2020.
-
- Das A, McGuire PG, Rangasamy S. Diabetic macular edema: Pathophysiology and novel therapeutic targets. Ophthalmology. 2015;122:1375–94. - PubMed
-
- Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin epidemiologic study of diabetic retinopathy. XV. The long-term incidence of macular edema. Ophthalmology. 1995;102:7–16. - PubMed
-
- Funatsu H, Yamashita H, Noma H, Mimura T, Yamashita T, Hori S. Increased levels of vascular endothelial growth factor and interleukin-6 in the aqueous humor of diabetics with macular edema. Am J Ophthalmol. 2002;133:70–7. - PubMed
-
- Brown DM, Nguyen QD, Marcus DM, Boyer DS, Patel S, Feiner L, et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: The 36-month results from two phase III trials: RISE and RIDE. Ophthalmology. 2013;120:2013–22. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

