Postprostatectomy Radiotherapy Timing and Long-Term Health-Related Quality of Life
- PMID: 39446326
- PMCID: PMC11581678
- DOI: 10.1001/jamanetworkopen.2024.40747
Postprostatectomy Radiotherapy Timing and Long-Term Health-Related Quality of Life
Abstract
Importance: The association between radiotherapy (RT) timing after radical prostatectomy and long-term patient-reported health-related quality of life (HRQOL) in men with prostate cancer is unknown.
Objective: To measure long-term HRQOL in men with prostate cancer up to 15 years after prostatectomy with or without RT and examine whether early vs late postprostatectomy RT is associated with differences in sexual, urinary, and bowel HRQOL.
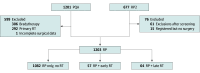
Design, setting, and participants: A prospective, multicenter, longitudinal cohort analysis using HRQOL data from the PROST-QA (2003-2006) and RP2 consortium (2010-2013) studies was conducted. Men with localized prostate cancer undergoing radical prostatectomy were included. Data were analyzed between May 8, 2023, and March 1, 2024. The study was conducted in 12 high-volume academic medical centers in the US.
Exposures: Men were stratified based on receipt and timing of postprostatectomy RT: prostatectomy only, early RT (<12 months), and late RT (≥12 months).
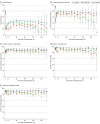
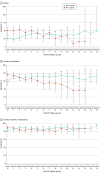
Main outcomes and measures: Longitudinal sexual, incontinence, urinary irritation, bowel, and hormonal/vitality HRQOL were measured via the Expanded Prostate Cancer Index Composite at baseline; months 2, 6, and 12; and annually thereafter. Treatment groups were compared using multivariable linear mixed-effects models of change in longitudinal domain scores. Pad use for incontinence was measured longitudinally among men receiving postprostatectomy RT.
Results: A total of 1203 men were included in the study: prostatectomy only (n = 1082), early RT (n = 57), and late RT (n = 64). Median age for the entire cohort was 60.5 (range, 38.8-79.7) years, and 1075 men (92.0%) were White. Median follow-up was 85.6 (IQR, 35.8-117.2) months. Compared with men receiving prostatectomy alone, those receiving postprostatectomy RT had significantly greater decreases in sexual, incontinence, and urinary irritation HRQOL. However, timing of postprostatectomy RT, specifically early vs late, was not associated with a long-term decrease in any HRQOL domain. There was evidence of improved recovery of sexual, continence, and urinary irritation scores among men receiving early RT compared with those receiving late RT after prostatectomy. Before the start of postprostatectomy RT, 39.3% of men in the early RT cohort and 73.4% of men in the late RT cohort were pad-free. By the sixth visit post-RT, 67.4% in the early RT cohort and 47.6% in the late RT cohort were pad-free.
Conclusions and relevance: In this multicenter, prospective analysis, postprostatectomy RT appeared to be negatively associated with long-term HRQOL across all domains. However, receipt of early vs late postprostatectomy RT may result in similar long-term HRQOL outcomes.
Conflict of interest statement
Figures
References
-
- Bolla M, van Poppel H, Tombal B, et al. ; European Organisation for Research and Treatment of Cancer, Radiation Oncology and Genito-Urinary Groups . Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: long-term results of a randomised controlled trial (EORTC trial 22911). Lancet. 2012;380(9858):2018-2027. doi: 10.1016/S0140-6736(12)61253-7 - DOI - PubMed
-
- Thompson IM, Tangen CM, Paradelo J, et al. Adjuvant radiotherapy for pathological T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: long-term followup of a randomized clinical trial. J Urol. 2009;181(3):956-962. doi: 10.1016/j.juro.2008.11.032 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

