A digital health-supported and community pharmacy-based lifestyle intervention program for adults with pre-diabetes: a study protocol for a cluster randomised controlled trial
- PMID: 39448216
- PMCID: PMC11579589
- DOI: 10.1136/bmjopen-2024-083921
A digital health-supported and community pharmacy-based lifestyle intervention program for adults with pre-diabetes: a study protocol for a cluster randomised controlled trial
Abstract
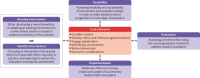
Introduction: Pre-diabetes indicates an elevated risk of developing type-2 diabetes and presents a window for preventive actions. The Pre-diabetes Intervention, Management and Evaluation (PRIME) programme is a community pharmacy-based pre-diabetes management programme that uses a mobile application for self-monitoring and pre-diabetes education, aiming to promote lifestyle changes among participants with pre-diabetes.
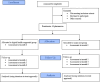
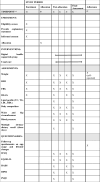
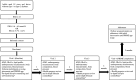
Methods and analysis: This is a protocol for a cluster randomised controlled trial that aims to evaluate the impact of the PRIME programme on participants' clinical outcomes and explore participants' and pharmacists' views towards its implementation. This protocol describes the development of the PRIME programme and mobile app, its feasibility and implementation in community pharmacy settings. 16 pharmacies from two states in Malaysia will be randomised to the intervention arm or standard care. The study will include overweight or obese adults with pre-diabetes. During each follow-up visit at the pharmacy, intervention participants will receive in-depth counselling from pharmacists after reviewing their self-monitoring data recorded in the PRIME app. They will also receive pre-diabetes education through the app and join a peer support chatgroup. The primary clinical outcome includes changes in body weight at 6 months, while the secondary clinical outcomes include changes in blood glucose profile, lipid profile, blood pressure and adiposity measures. The sustainability of the PRIME programme will be accessed using a follow-up questionnaire, while participants' engagement with the intervention will be evaluated using attendance rate and the app data. Focus group discussions and one-to-one interviews will be conducted for process evaluation. This study will inform the impact of community pharmacists-led digital health intervention in pre-diabetes management.
Ethics and dissemination: This study has been registered with clinicaltrials.gov (NCT04832984) and approved by the Monash University Human Research Ethics Committee (Project ID: 27512).
Trial registration number: clinicaltrials.gov (NCT04832984).
Keywords: Behaviour; Clinical trials; Diabetes & Endocrinology; Epidemiology; Primary Prevention; eHealth.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures
References
-
- International Diabetes Federation . IDF Diabetes Atlas. 10th. 2021. edn.
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
