Investigation of the Effect of High Shear Stress on Mesenchymal Stem Cells Using a Rotational Rheometer in a Small-Angle Cone-Plate Configuration
- PMID: 39451387
- PMCID: PMC11504001
- DOI: 10.3390/bioengineering11101011
Investigation of the Effect of High Shear Stress on Mesenchymal Stem Cells Using a Rotational Rheometer in a Small-Angle Cone-Plate Configuration
Abstract
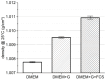
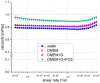
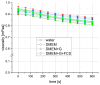
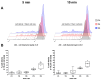
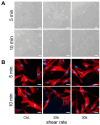
Within the healthy human body, cells reside within the physiological environment of a tissue compound. Here, they are subject to constant low levels of mechanical stress that can influence the growth and differentiation of the cells. The liposuction of adipose tissue and the subsequent isolation of mesenchymal stem/stromal cells (MSCs), for example, are procedures that induce a high level of mechanical shear stress. As MSCs play a central role in tissue regeneration by migrating into regenerating areas and driving regeneration through proliferation and tissue-specific differentiation, they are increasingly used in therapeutic applications. Consequently, there is a strong interest in investigating the effects of shear stress on MSCs. In this study, we present a set-up for applying high shear rates to cells based on a rotational rheometer with a small-angle cone-plate configuration. This set-up was used to investigate the effect of various shear stresses on human adipose-derived MSCs in suspension. The results of the study show that the viability of the cells remained unaffected up to 18.38 Pa for an exposure time of 5 min. However, it was observed that intense shear stress damaged the cells, with longer treatment durations increasing the percentage of cell debris.
Keywords: cone and plate configuration; high shear rates; low viscous liquids; mesenchymal stem/stromal cells; rotational rheometer; thin-gap rheometry.
Conflict of interest statement
Author Juliane Meyer was employed by the Human Med AG. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Rigotti G., Marchi A., Galiè M., Baroni G., Benati D., Krampera M., Pasini A., Sbarbati A. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: A healing process mediated by adipose-derived adult stem cells. Plast. Reconstr. Surg. 2007;119:1409–1422. doi: 10.1097/01.prs.0000256047.47909.71. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

