Design and antiviral assessment of a panel of fusion proteins targeting human papillomavirus type 16
- PMID: 39453911
- PMCID: PMC11508125
- DOI: 10.1371/journal.pone.0311137
Design and antiviral assessment of a panel of fusion proteins targeting human papillomavirus type 16
Abstract
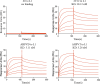
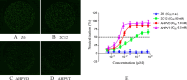
Cervical cancer ranks as the third most prevalent malignancy in women worldwide. The persistence of Human papillomavirus (HPV) infection stands out as the foremost risk factor for cervical cancer development. Among the numerous HPV subtypes, HPV16 infection emerges as the primary pathogenic determinant of cervical cancer. To date, no specific drugs have been approved. In this study, we engineered two high-affinity fusion protein targeting HPV16 L1 protein based on the alpaca-derived single-domain antibody 2C12 previously obtained in our laboratory. These two fusion proteins exhibited potent neutralizing activity against HPV16 pseudovirus with IC50 values of 7.8 nM and 6.5 nM, respectively. Molecular docking analysis revealed that 2C12 formed ten pairs of hydrogen bonds with HPV16 L1, among which Arg39 and Thr100 established multiple pairs of hydrogen bonds with HPV16 L1, indicating their crucial roles in antigen-antibody binding process. These structural and biological findings underscore the effective binding capacity of these fusion proteins to HPV16, leading to reduced viral load and providing valuable insights into therapeutic antibody and vaccine development against HPV 16 infection.
Copyright: © 2024 Bai et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

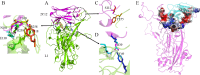
Similar articles
-
Human papillomavirus 16 L1-E7 chimeric virus like particles show prophylactic and therapeutic efficacy in murine model of cervical cancer.Vaccine. 2012 Aug 3;30(36):5417-24. doi: 10.1016/j.vaccine.2012.06.010. Epub 2012 Jun 17. Vaccine. 2012. PMID: 22717329
-
Developments in L2-based human papillomavirus (HPV) vaccines.Virus Res. 2017 Mar 2;231:166-175. doi: 10.1016/j.virusres.2016.11.020. Epub 2016 Nov 23. Virus Res. 2017. PMID: 27889616 Free PMC article. Review.
-
Vaccination of healthy volunteers with human papillomavirus type 16 L2E7E6 fusion protein induces serum antibody that neutralizes across papillomavirus species.Cancer Res. 2006 Dec 1;66(23):11120-4. doi: 10.1158/0008-5472.CAN-06-2560. Cancer Res. 2006. PMID: 17145854
-
Correlation between cervical HPV DNA detection and HPV16 seroreactivity measured with L1-only and L1+L2 viral capsid antigens.J Med Microbiol. 2020 Jul;69(7):960-970. doi: 10.1099/jmm.0.001213. J Med Microbiol. 2020. PMID: 32510304 Free PMC article.
-
Worldwide genetic variations in high-risk human papillomaviruses capsid L1 gene and their impact on vaccine efficiency.Gene. 2021 May 25;782:145533. doi: 10.1016/j.gene.2021.145533. Epub 2021 Feb 23. Gene. 2021. PMID: 33636291 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

