A humanized monoclonal antibody targeting an ectonucleotidase rescues cardiac metabolism and heart function after myocardial infarction
- PMID: 39454569
- PMCID: PMC11604407
- DOI: 10.1016/j.xcrm.2024.101795
A humanized monoclonal antibody targeting an ectonucleotidase rescues cardiac metabolism and heart function after myocardial infarction
Abstract
Myocardial infarction (MI) results in aberrant cardiac metabolism, but no therapeutics have been designed to target cardiac metabolism to enhance heart repair. We engineer a humanized monoclonal antibody against the ectonucleotidase ENPP1 (hENPP1mAb) that targets metabolic crosstalk in the infarcted heart. In mice expressing human ENPP1, systemic administration of hENPP1mAb metabolically reprograms myocytes and non-myocytes and leads to a significant rescue of post-MI heart dysfunction. Using metabolomics, single-nuclear transcriptomics, and cellular respiration studies, we show that the administration of the hENPP1mAb induces organ-wide metabolic and transcriptional reprogramming of the heart that enhances myocyte cellular respiration and decreases cell death and fibrosis in the infarcted heart. Biodistribution and safety studies showed specific organ-wide distribution with the antibody being well tolerated. In humanized animals, with drug clearance kinetics similar to humans, we demonstrate that a single "shot" of the hENPP1mAb after MI is sufficient to rescue cardiac dysfunction.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The intellectual property associated with hENPP1mAb is held by the Regents, University of California.
Figures






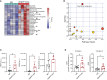
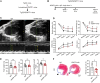
References
-
- Gulati A., Jabbour A., Ismail T.F., Guha K., Khwaja J., Raza S., Morarji K., Brown T.D.H., Ismail N.A., Dweck M.R., et al. Association of fibrosis with mortality and sudden cardiac death in patients with nonischemic dilated cardiomyopathy. JAMA, J. Am. Med. Assoc. 2013;309:896–908. doi: 10.1001/jama.2013.1363. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

