Chemical and Biological Characterization of Metabolites from Silene viridiflora Using Mass Spectrometric and Cell-Based Assays
- PMID: 39456218
- PMCID: PMC11505650
- DOI: 10.3390/biom14101285
Chemical and Biological Characterization of Metabolites from Silene viridiflora Using Mass Spectrometric and Cell-Based Assays
Abstract
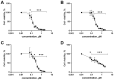
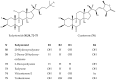
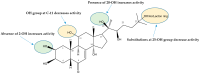
A comprehensive metabolite profiling of the medicinal plant Silene viridiflora using an UHPLC-ESI-MS/MS method is described for the first time. A total of 71 compounds were identified and annotated, the most common of which were flavonoids, triterpene glycosides, and ecdysteroids. The three major compounds schaftoside, 26-hydroxyecdysone, and silviridoside can be chosen as the markers for the assessment of the quality of S. viridiflora preparations. The methanol extract and a variety of metabolites identified in S. viridiflora were screened for their cytotoxic and Wnt pathway-inhibiting activities against triple-negative breast cancer (TNBC), the deadliest form of cancer in women. 2-Deoxy-20-hydroxyecdysone with submicromolar IC50 was identified as a result. The structure-activity relationship derived from the data from the in vitro proliferation assay showed that the hydroxyl group present at position C-2 of steroid core reduces the ecdysteroids' cytotoxicity against cancer cells.
Keywords: Silene viridiflora; TNBC; UHPLC-MS; Wnt signaling; cytotoxicity; metabolite annotation.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Phytoecdysteroids of Silene guntensis and their in vitro cytotoxic and antioxidant activity.Z Naturforsch C J Biosci. 2011 May-Jun;66(5-6):215-24. doi: 10.1515/znc-2011-5-604. Z Naturforsch C J Biosci. 2011. PMID: 21812338
-
Silviridoside: A New Triterpene Glycoside from Silene viridiflora with Promising Antioxidant and Enzyme Inhibitory Potential.Molecules. 2022 Dec 11;27(24):8781. doi: 10.3390/molecules27248781. Molecules. 2022. PMID: 36557914 Free PMC article.
-
Comparative metabolic profiling and quantitative analysis of metabolites in different tissues of Ajuga turkestanica by ESI-UHPLC-QqTOF-MS and NMR.Sci Rep. 2024 Nov 15;14(1):28179. doi: 10.1038/s41598-024-71546-5. Sci Rep. 2024. PMID: 39548128 Free PMC article.
-
The Natural Products and Extracts: Anti-Triple-Negative Breast Cancer in Vitro.Chem Biodivers. 2021 Jul;18(7):e2001047. doi: 10.1002/cbdv.202001047. Epub 2021 Jun 4. Chem Biodivers. 2021. PMID: 34000082 Review.
-
The Potential of Flavonoids and Tannins from Medicinal Plants as Anticancer Agents.Anticancer Agents Med Chem. 2020;20(18):2216-2227. doi: 10.2174/1871520620666200516150829. Anticancer Agents Med Chem. 2020. PMID: 32416704 Review.
Cited by
-
Success and Controversy of Natural Products as Therapeutic Modulators of Wnt Signaling and Its Interplay with Oxidative Stress: Comprehensive Review Across Compound Classes and Experimental Systems.Antioxidants (Basel). 2025 May 14;14(5):591. doi: 10.3390/antiox14050591. Antioxidants (Basel). 2025. PMID: 40427472 Free PMC article. Review.
References
-
- Jakimiuk K., Wink M., Tomczyk M. Flavonoids of the Caryophyllaceae. Phytochem. Rev. 2022;21:179–218. doi: 10.1007/s11101-021-09755-3. - DOI
-
- Mamadalieva N.Z., Lafont R., Wink M. Diversity of secondary metabolites in the genus Silene L. (Caryophyllaceae)—Structures, Distribution, and Biological properties. Diversity. 2014;6:415–499. doi: 10.3390/d6030415. - DOI
-
- Makhmudova M.M., Bacher M., Zengin G., Rosenau T., Youssef F.S., Almasri D.M., Elhady S.S., Mamadalieva N.Z. Silviridoside: A New Triterpene Glycoside from Silene viridiflora with Promising Antioxidant and Enzyme Inhibitory Potential. Molecules. 2022;27:8781. doi: 10.3390/molecules27248781. - DOI - PMC - PubMed
-
- Dzakhangirova M., Syrov V. Experimental evaluation of effect of stimulation of sum of ecdysteroids from Silene brachuica and S. viridiflora for erythropoiesis in laboratory animals. Pathology. 2005;2:7–9.
-
- Shakhmurova G.A., Mamadalieva N.Z., Zhanibekov A.A., Khushbaktova Z.A., Syrov V.N. Effect of total ecdysteroid preparation from Silene viridiflora on the immune state of experimental animals under normal and secondary immunodeficiency conditions. Pharm. Chem. J. 2012;46:222–224. doi: 10.1007/s11094-012-0766-4. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

