An Overview of the Mechanisms through Which Plants Regulate ROS Homeostasis under Cadmium Stress
- PMID: 39456428
- PMCID: PMC11505430
- DOI: 10.3390/antiox13101174
An Overview of the Mechanisms through Which Plants Regulate ROS Homeostasis under Cadmium Stress
Abstract
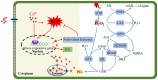
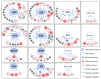
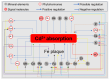
Cadmium (Cd2+) is a non-essential and highly toxic element to all organic life forms, including plants and humans. In response to Cd stress, plants have evolved multiple protective mechanisms, such as Cd2+ chelation, vesicle sequestration, the regulation of Cd2+ uptake, and enhanced antioxidant defenses. When Cd2+ accumulates in plants to a certain level, it triggers a burst of reactive oxygen species (ROS), leading to chlorosis, growth retardation, and potentially death. To counteract this, plants utilize a complex network of enzymatic and non-enzymatic antioxidant systems to manage ROS and protect cells from oxidative damage. This review systematically summarizes how various elements, including nitrogen, phosphorus, calcium, iron, and zinc, as well as phytohormones such as abscisic acid, auxin, brassinosteroids, and ethylene, and signaling molecules like nitric oxide, hydrogen peroxide, and hydrogen sulfide, regulate the antioxidant system under Cd stress. Furthermore, it explores the mechanisms by which exogenous regulators can enhance the antioxidant capacity and mitigate Cd toxicity.
Keywords: antioxidant enzymes; antioxidants; cadmium; reactive oxygen species; regulatory mechanism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Shah A.A., Aslam S., Akbar M., Ahmad A., Khan W.U., Yasin N.A., Ali B., Rizwan M., Ali S. Combined effect of Bacillus fortis IAGS 223 and zinc oxide nanoparticles to alleviate cadmium phytotoxicity in Cucumis melo. Plant Physiol. Biochem. 2021;158:1–12. doi: 10.1016/j.plaphy.2020.11.011. - DOI - PubMed
Publication types
Grants and funding
- ISSAS2412/Field Frontier Program of the Institute of Soil Science
- CX(22)3138/Jiangsu Agricultural Science and Technology Innovation Fund
- ZSBBL-KY2023-07/Zhongshan Biological Breeding Laboratory
- 2023kj01/Yafu Technology Innovation and Service Major Project of Jiangsu Vocational College of Agriculture and Forestry
- 2021kj15/Fund Support Project of Jiangsu Vocational College of Agriculture and Forestry
LinkOut - more resources
Full Text Sources

