Oncolytic Coxsackievirus B3 Strain PD-H Is Effective Against a Broad Spectrum of Pancreatic Cancer Cell Lines and Induces a Growth Delay in Pancreatic KPC Cell Tumors In Vivo
- PMID: 39457005
- PMCID: PMC11508574
- DOI: 10.3390/ijms252011224
Oncolytic Coxsackievirus B3 Strain PD-H Is Effective Against a Broad Spectrum of Pancreatic Cancer Cell Lines and Induces a Growth Delay in Pancreatic KPC Cell Tumors In Vivo
Abstract
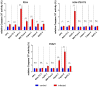
Pancreatic cancer is one of the deadliest cancers globally, with limited success from existing therapies, including chemotherapies and immunotherapies like checkpoint inhibitors for patients with advanced pancreatic ductal adenocarcinoma (PDAC). A promising new approach is the use of oncolytic viruses (OV), a form of immunotherapy that has been demonstrated clinical effectiveness in various cancers. Here we investigated the potential of the oncolytic coxsackievirus B3 strain (CVB3) PD-H as a new treatment for pancreatic cancer. In vitro, PD-H exhibited robust replication, as measured by plaque assays, and potent lytic activity, as assessed by XTT assays, in most pancreatic tumor cell lines, outperforming two other coxsackievirus strains tested, H3N-375/1TS and CVA21. Thus, H3N-375/1TS showed efficient replication and lytic efficiency in distinctly fewer tumor cell lines, while most tumor cells were resistant to CVA21. The oncolytic efficiency of the three OV largely correlated with mRNA expression levels of viral receptors and their ability to induce apoptosis, as measured by cleaved caspase 3/7 activity in the tumor cells. In a syngeneic mouse model with subcutaneous pancreatic tumors, intratumoral administration of PD-H significantly inhibited tumor growth but did not completely stop tumor progression. Importantly, no virus-related side effects were observed. Although pancreatic tumors respond to PD-H treatment, its therapeutic efficacy is limited. Combining PD-H with other treatments, such as those aiming at reducing the desmoplastic stroma which impedes viral infection and spread within the tumor, may enhance its efficacy.
Keywords: CVA21; KPC cells; PD-H; coxsackievirus; oncolytic virus; pancreatic cancer.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





References
-
- Neoptolemos J.P., Stocken D.D., Bassi C., Ghaneh P., Cunningham D., Goldstein D., Padbury R., Moore M.J., Gallinger S., Mariette C., et al. Adjuvant chemotherapy with fluorouracil plus folinic acid vs gemcitabine following pancreatic cancer resection: A randomized controlled trial. J. Am. Med. Assoc. 2010;304:1073–1081. doi: 10.1001/jama.2010.1275. - DOI - PubMed
-
- Burris H.A., Moore M.J., Andersen J., Green M.R., Rothenberg M.L., Modiano M.R., Cripps M.C., Portenoy R.K., Storniolo A.M., Tarassoff P., et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997;15:2403–2413. doi: 10.1200/JCO.1997.15.6.2403. - DOI - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

